Publications
There have been a number of articles published in support of Corsano’s and its partners' scientific approach.The accuracy of heartbeat detection using photoplethysmography technology in cardiac patients
This validation study showed that the Corsano 287 CardioWatch/Bracelet with PPG-technology can determine HR and RR-intervals with high accuracy in a cardiovascular patient population, with high quality output in different subgroups, especially when combined with a signal quality indicator. Due to their non-intrusive and convenient nature, wearable devices like these have great potential for high volume accessible long-term monitoring at-risk cardiac patients.
S. Blok 1,2*, M.A. Piek 1, I.I. Tulevski 1, G.A. Somsen 1, M.M. Winter 1,3
- Department of Cardiology, Cardiology Centers of the Netherlands, Karel du Jardinstraat 61, Amsterdam, the Netherlands
- Department of Vascular Medicine, Internal Medicine, Amsterdam UMC, University of Amsterdam, Amsterdam Cardiovascular Sciences, Meibergdreef 9, Amsterdam, the Netherlands
- Department of Cardiology, Amsterdam UMC, University of Amsterdam, Amsterdam Cardiovascular Sciences, Meibergdreef 9, Amsterdam, the Netherlands
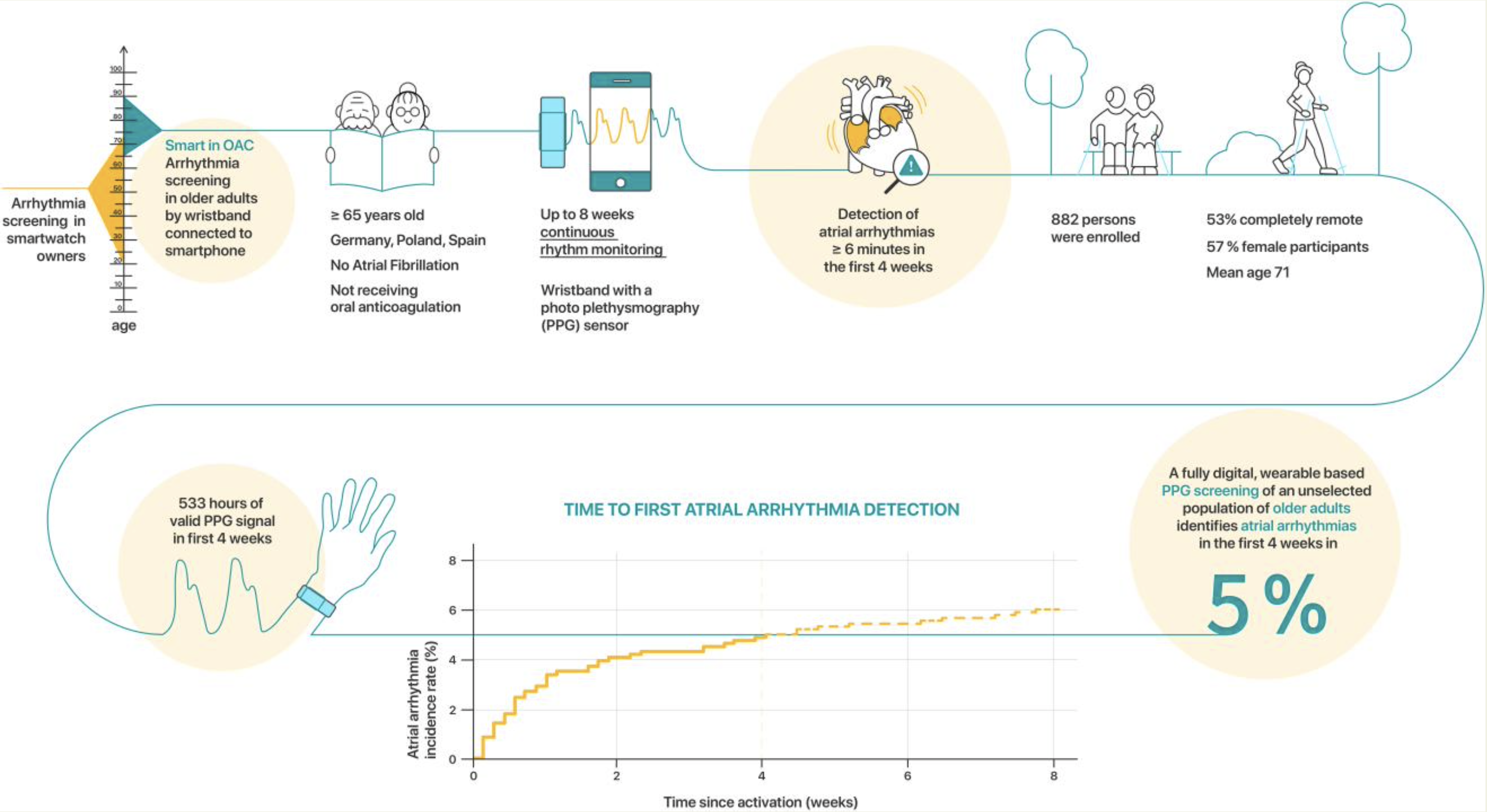
Smartphone and wearable detected atrial arrhythmias in Older Adults: Results of a fully digital European Case finding study
The fully remote, investigator-initiated Smartphone and Corsano Health’s CardioWatch 287 wearable detected atrial arrhythmia in Older Adults Case finding study (Smart in OAC—AFNET 9) digitally enrolled participants ≥65 years without known atrial fibrillation, not receiving oral anticoagulation in Germany, Poland, and Spain for 8 weeks. Participants were invited by media communications and direct contacts. Study procedures adhered to European data protection. Consenting participants received a wristband with a photoplethysmography sensor to be coupled to their smartphone. The primary outcome was the detection of atrial arrhythmias lasting 6 min or longer in the first 4 weeks of monitoring.
Conclusion: Remote, digitally supported consumer-electronics-based screening is feasible in older European adults and identifies atrial arrhythmias in 5% of participants within 4 weeks of monitoring.
L Fabritz 1,2,3,4,5 *, DL Connolly 4,6, E Czarnecki 5, D Dudek 7,8, E Guasch 9,10,11, D Haase 5, T Huebner 12, A Zlahoda-Huzior 13, K Jolly 14, P Kirchhof 2,3,4,5, J Obergassel 1,2,3, U Schotten 5,15, E Vettorazzi 16, SJ Winkelmann 1,2, A Zapf 16, and RB Schnabel 2,3,5
- University Center of Cardiovascular Science, University Heart and Vascular Center Hamburg, University Medical Center Hamburg-Eppendorf, Martinistr. 52, 20251 Hamburg, Germany
- Department of Cardiology, University Heart and Vascular Center Hamburg, Martinistr. 52, 20251 Hamburg, Germany
- DZHK German Center for Cardiovascular Research,
partner site Hamburg/Luebeck/Kiel, Germany - Institute of Cardiovascular Sciences, University of Birmingham, Edgbaston Wolfson Drive, B15 2TT Birmingham, UK
- Atrial Fibrillation NETwork (AFNET), Mendelstr 11, 48149 Münster, Germany
- Department of Cardiology and R&D, Birmingham City Hospital, Sandwell and West Birmingham Trust, Dudley Road, B18 7QH Birmingham, UK
- Jagiellonian University Medical College, Center for Digital Medicine and Robotics, Ul. Kopernika 7E, 33-332 Kraków, Poland
- Maria Cecilia Hospital, Via Corriera, 1, 48033 Cotignola RA, Italy
- Institut Clínic Cardio-Vascular, Hospital Clínic, University of Barcelona, Carrer de Villaroel, 170, 08036 Barcelona, CA, Spain, Spain
- IDIBAPS, Rosselló 149-153, 08036 Barcelona, CA, Spain
- CIBERCV, Monforte de Lemos 3-5, Pabellon 11, Planta 0, 28029 Madrid, Spain
- Preventicus GmbH, Ernst-Abbe-Straße 15, 07743
Jena, Germany - Department of Measurement and Electronics, AGH University of Science and Technology, Al. Mickiewicza 30, 30-059 Kraków, Poland
- Institute of Applied Health Research, University of Birmingham, Edgbaston, B15 2TT Birmingham, UK
- Department of Physiology, Cardiovascular Research Institute Maastricht, Maastricht University Medical
Center +, Debyelaan 25, 6229 HX, Maastricht, The Netherlands - Institute of Medical Biometry and Epidemiology, University Medical Center Hamburg-Eppendorf, ChristophProbst-Weg 1, 20246 Hamburg, Germany
European Heart Journal - Digital Health, Volume 3, Issue 4, December 2022, Pages 610-625, https://doi.org/10.1093/ehjdh/ztac067
Validation of a novel cuffless photoplethysmography-based wristband for measuring blood pressure according to the regulatory standard
Background: Chronic hypertension is a significant risk factor for cardiovascular diseases (CVD) [1]. However, obtaining reliable and reproducible cuff blood pressure (BP) measurements is challenging. We demonstrate that a wristband-based PPG measurement in combination with a custom developed BP algorithm represents a possible alternative to cuff BP measurements.
Purpose: This study aimed to validate a novel cuffless photopletysmography (PPG)-based non-invasive wristband for continuous BP monitoring in accordance with ISO 81060-2:2019.
Methods: The study compared PPG-guided BP algorithm predictions with subclavian arterial reference measurements taken during cardiac catheterization. Eligible patients were consecutively included, and eligibility was screened following ISO 81060-2:2019 requirements. Reference measurements were performed using a validated invasive BP monitoring device with a sampling rate of 100Hz. PPG signals were collected using six light emission diodes and two photodiodes at a sampling rate of 128Hz. Three sequential initialization measurements were taken using a validated blood pressure cuff before the cardiac catheterization exam. These measurements, along with approximately 100 additional features (PPG-derived and based on patient demographics), were used as input for the machine learning-based BP algorithm. Correlation, mean error, and standard deviation (SD) were determined for systolic and diastolic BP measurements between the BP algorithm predictions and invasive reference measurements
Results: The study included 97 patients from whom 420 individual 30-second samples were obtained. The mean age, weight, and height of the analysed subjects were 67.1 (SD 11.1), 83.4 (SD 16.1), and 174.1 (SD 10.0), respectively. In 48 samples (11%), systolic BP was ≤100mmHg, while in 106 samples (25%), systolic BP was ≥160mmHg. Diastolic BP was ≤70 mmHg in 222 samples (53%) and ≥85 mmHg in 99 samples (24%). The BP algorithm predictions showed a high correlation with invasive reference measurements for systolic (r = 0.985) and diastolic (r = 0.961) BP measurements. The mean error of the BP algorithm predictions compared to the invasive reference measurements was ±3.7 mmHg (SD 4.4 mmHg) and ±2.5 mmHg (SD 3.7 mmHg) for systolic and diastolic BP, respectively. Results were similar within each gender and skin colour category (Fitzpatrick I-VI).
Conclusion: This study demonstrates that a wristband-based PPG measurement in combination with the developed BP algorithm can provide accurate continuous BP measurements across a wide range of BP distributions. Therefore, wristband BP monitoring may serve as a valid and less burdensome alternative to cuff BP measurements for both in-hospital and at-home BP monitoring. However, further research is necessary to evaluate the precision of the BP algorithm during movement and the stability of the predictions over time.
Eelko Ronner (ER), MD, PhD 1; Mariska van Vliet, MD, PhD 1; Stefan H.J. Monnink, MD, PhD 1; Mathijs J. Kuiper, MD 1; Dieke Hoftijzer, MD, PhD 1; Jan C. Constandse, MD 1
- Department of Cardiology, Reinier de Graaf Hospital, Reinier de Graafweg 5, Delft, The Netherlands.
Dr. Eelko Ronner PhD presented study findings Cuffless Non-Invasive Blood Pressure at American Heart Association 2023 Scientific Sessions.
https://www.ahajournals.org/doi/10.1161/circ.148.suppl_1.17703
Automated cardiac arrest DETECTion based on wrist-derived PPG signals: preliminary results of the DETECT-1 study in patients with induced circulatory arrests (Abstract)
Automated cardiac arrest detection and alarming is a potential solution to shorten treatment delays after out-of-hospital cardiac arrest (OHCA). Especially for patients with unwitnessed cardiac arrests this could improve survival. In the DETECT project, an existing wristband is further developed to enable automated cardiac arrest detection and alarming during daily life using multiple sensors including wrist-derived photoplethysmography (PPG) signals. We present preliminary results of the DETECT-1 study in which a PPG-based cardiac arrest detection algorithm is being developed using patient data of induced circulatory arrests. (Abstract)
Edgar R.1, Scholte N.2, Ebrahimkheil K.3, Brouwer M.1, Beukema R.1, van Mieghem N.2, Yap S. 2, Rodwell L. 4, Ronner E. 5, Boersma E. 2, Stas P.C.3, van Royen N.1, Bonnes J.L.1
- Department of Cardiology, Radboud University Medical Center, Nijmegen, the Netherlands
- Department of Cardiology, Erasmus Medical Center, Rotterdam, the Netherlands
- Corsano Health, The Hague, the Netherlands
- Radboud Institute for Health Sciences, Health Evidence, Section Biostatistics, Nijmegen, the Netherlands
- Department of Cardiology, Reinier de Graaf hospital , Delft, the Netherlands
Determine atrial fibrillation burden with a photoplethysmographic mobile sensor: the atrial fibrillation burden trial: detection and quantification of episodes of atrial fibrillation using a cloud analytics service connected to a wearable with photoplethysmographic sensor
Recent studies suggest that atrial fibrillation (AF) burden (time AF is present) is an independent risk factor for stroke. The aim of this trial was to study the feasibility and accuracy to identify AF episodes and quantify AF burden in patients with a known history of paroxysmal AF with a photoplethysmography (PPG)-based CardioWatch 287.
European Heart Journal - Digital Health, ztad039, https://doi.org/10.1093/ehjdh/ztad039
Pamela Reissenberger 1, Peter Serfözö 1, Diana Piper 2, Norman Juchler 3, Sara Glanzmann 1, Jasmin Gram 1, Karina Hensler 1, Hannah Tonidandel 1, Elena Börlin 4, Marcus D’Souza 4,5, Patrick Badertscher 6, and Jens Eckstein 1,4
- Department of Internal Medicine, University Hospital Basel, Petersgraben 4, 4031 Basel, Switzerland
- Preventicus, Ernst-Abbe-Str. 15, 07743 Jena, Germany
- Institute of Computational Life Sciences, Zurich University of Applied Sciences, Schloss 1, 8820 Wädenswil, Switzerland
- Department Digitalization & ICT, University Hospital Basel, Spitalstrasse 26, 4031 Basel, Switzerland
- Department of Neurology, University Hospital Basel, Petersgraben 4, 4031 Basel, Switzerland
- Department of Cardiology, University Hospital Basel, Petersgraben 4, 4031 Basel, Switzerland
Continuous respiration rate monitoring using photoplethysmography technology in patients with Obstructive Sleep Apnea
Respiration rate is an important physiological parameter whose abnormality has been regarded as an important indicator of various serious illnesses. Photoplethysmography (PPG) in wearable sensors potentially plays an important role in early disease detection by making respiration rate measurements more accessible. We investigated the accuracy of a new non-invasive, continuous, wrist-worn and wireless monitoring PPG device (Corsano CardioWatch 287) in measuring respiration rate (RR) and pulse rate (PR) at rest.
J.M. Gehring 1, E.J. Japenga 1,2, L.C. Saeijs-van Niel 1, L.P ten Bosch-Paniagua 1, M.H. Frank 1,3
1. Department of Sleep Medicine, Haaglanden Clinics, Nieuwe Parklaan 11, The Hague, The Netherlands
2. Department of Respiratory Medicine, Haaglanden Medisch Centrum, Lijnbaan 32, The Hague, The Netherlands
3. Department of Oral Surgery, Haaglanden Clinics, Nieuwe Parklaan 11, The Hague, The Netherlands
medRxiv Yale University, and BMJ
https://doi.org/10.1101/2023.07.25.23293134
Validation Corsano Steps Algorithm (Abstract)
Comparison measured steps per minute determined by the Cardiowatch with steps per minute determined with the Actigraph CENTREPOINT Insight Watch. Single center prospective observational study collecting PPG and accelerometer data in a real world setting. For the individual participants, study duration was between 1 and 5 days, depending on their preference. PPG and accelerometer data were collected during normal daily activities performed, including skiing.
Methodology: To assess the Cardiowatch step counting accuracy based on a custom Corsano Health algorithm. To do so the amount of steps per minute determined by the CW2 using both algorithms were compared to the amount of steps determined by CE/FDA certified Actigraph CENTREPOINT Insight Watch.
Measurements were performed in 14 adult healthy volunteers of the cardiology and cardiothoracic surgery department of the Radboudumc. After obtaining informed consent, participants wore the Cardiowatch 287-2 wristband (equipped with an accelerometer and PPG sensors) on the left or right wrist. Reference measurements were performed using the Actigraph watch on the opposite wrist.
Results: Overview of statistical analysis of amount of counted steps by Cardiowatch 287-2 using the Corsano Health algorithm compared to the Actigraph reference.
|
RMSE
|
1.2 |
|
Bias (+95% CI)
|
0.7 (0.6, 0.7) |
|
95% LoA (+ 95% CI) Lower |
-2.5 (-2.5, -2.4) |
|
95% LoA (+ 95% CI) Upper |
3.8 (3.7, 3.8) |
Edgar R.1, Ebrahimkheil K. 2, Bonnes J.L.1
1. Department of Cardiology, Radboud University Medical Center, Nijmegen, the Netherlands
2. Corsano Health, The Hague, the Netherlands

Analyzability of Photoplethysmographic Smartwatch Data During Everyday Life: Feasibility Study
A total of 41 (19 female and 22 male) adults in good cardiovascular health (aged 19-79 years) continuously wore a smartwatch equipped with a PPG sensor and a 3D accelerometer (Cardio Watch 287, Corsano Health BV) for a period of 24 hours that represented their individual daily routine. For each participant, smartwatch data were analyzed on a 1-minute basis by an algorithm designed for heart rhythm analysis (Preventicus Heartbeats, Preventicus GmbH). As outcomes, the percentage of analyzable data (PAD) and the mean acceleration (ACC) were calculated. To map changes of the ACC and PAD over the course of one day, the 24-hour period was divided into 8 subintervals comprising 3 hours each.
Steve Merschel, Diplom FH 1, Lars Reinhardt, PhD 2
- Preventicus GmbH, Jena, Germany
- Institute for Applied Training Science, Leipzig, Germany
Corsano Health Cardiowatch 287 USA Usability Study
Regulations and standards put the emphasis on usability and human factor design in the development of medical device and risk management. This paper explains the main challenges for wearable devices by showing how usability and human factors study was performed on the Corsano Cardiowatch 287 bracelet for body metrics monitoring.
Dr. Baran Erdik 1, Philippe Fraboulet 2
- Physician (MD / MHPA), Healthcare Compliance Expert
- Regulatory Expert Manufacture Modules Technologies
Remote Design of a Smartphone and Wearable Detected Atrial Arrhythmia in Older Adults Case Finding Study: Smart in OAC – AFNET 9
Screening for atrial fibrillation and timely initiation of oral anticoagulation, rhythm management, and treatment of concomitant cardiovascular conditions can improve outcomes in high-risk populations.
The multicenter, international, investigator-initiated, single-arm case-finding Smartphone and wearable detected atrial arrhythmia in older adults case finding study (Smart in OAC – AFNET 9) evaluates the diagnostic yield of a validated, cloud-based analysis algorithm detecting atrial arrhythmias via a signal acquired by a smartphone-coupled with Corsano Health’s CardioWatch 287 wristband monitoring system in older adults. Unselected participants aged ≥65 years without known atrial fibrillation and not receiving oral anticoagulation are enrolled in three European countries. Participants undergo continuous pulse monitoring using a wristband with a photo plethysmography (PPG) sensor and a telecare analytic service. Participants with PPG-detected atrial arrhythmias will be offered ECG loop monitoring. The study has a virtual design with digital consent and teleconsultations, whilst including hybrid solutions. Primary outcome is the proportion of older adults with newly detected atrial arrhythmias.
L Fabritz 1,2,3,4 *, DL Connolly 4,5, E Czarnecki 6, D Dudek 7,8, A Zlahoda-Huzior 9, E Guasch 10,11,12, D Haase 6, T Huebner 13, , K Jolly 14, P Kirchhof 2,3,4,6, U Schotten 6,15, A Zapf 16, and RB Schnabel 2,3,6
1. University Center of Cardiovascular Science, University Medical Center Hamburg-Eppendorf, Hamburg, Germany
2. Department of Cardiology, University Heart and Vascular Center Hamburg, University Medical Center Hamburg-Eppendorf, Hamburg, Germany
3. German Center for Cardiovascular Research, Partner Site Hamburg/Luebeck/Kiel, Berlin, Germany
4. Institute of Cardiovascular Sciences, University of Birmingham, Birmingham, United Kingdom
5. Department of Cardiology and R&D, Birmingham City Hospital, Sandwell and West Birmingham Trust, Birmingham, United Kingdom
6. Atrial Fibrillation NETwork, Münster, Germany
7. Institute of Cardiology, Jagiellonian University Medical College, Krakow, Poland
8. Maria Cecilia Hospital, GVM Care & Research, Ravennna, Italy
9. Department of Measurement and Electronics, AGH University of Science and Technology, Krakow, Poland
10. Institut Clínic Cardio-Vascular, Hospital Clínic, University of Barcelona, Barcelona, Spain
11. August Pi i Sunyer Biomedical Research Institute, Barcelona, Spain
12. Centro de Investigación Biomédica en Red en Enfermedades Cardiovasculares, Madrid, Spain
13. Preventicus GmbH, Jena, Germany
14. Institute of Applied Health Research, University of Birmingham, Birmingham, United Kingdom
15. Department of Physiology, Cardiovascular Research Institute Maastricht, Maastricht University Medical Center, Maastricht, Netherlands
16. Institute of Medical Biometry and Epidemiology, University Medical Center Hamburg-Eppendorf, Hamburg, Germany
Front. Cardiovasc. Med., 21 March 2022. Sec. Cardiac Rhythmology
Volume 9 - 2022 |https://doi.org/10.3389/fcvm.2022.839202
AF detection using Smartphone Apps
70 patients in an AF rhythm and 70 patients in sinus rhythm were recruited. Mean age was 73±12 (60% male) in the AF group, 59±20 in the sinus rhythm group (66% male). The BeatScanner app had a sensitivity of 89% (95% CI: 79-95%) and specificity of 67% (55- 78%). The Preventicus app had a sensitivity of 94% (86- 98%) and specificity of 96% (88-99%). There were 83/140 (59%) regular smartphone users, among whom 94% were interested in using their smartphone to self-screen for AF and 55% would pay for an app to do so. Almost all (96%) found the apps easy to use and 63% preferred the Preventicus app to the BeatScanner app. In a selected sample of hospital patients, the Preventicus app was more accurate for discriminating between AF and sinus rhythm than the BeatScanner app and was preferred by patients. Using the smartphones intrinsic function is a promising, simple and acceptable means of self-screening for AF
N. Karim 1; M. Aral 1; S. Eduawor 1; M. Finlay 1
1. Barts Heart Centre, London, United Kingdom
EP Europace, Volume 19, Issue suppl_3, June 2017, Page iii234
Validation of Photoplethysmography-Based Sleep Staging Compared With Polysomnography
Corsano’s sleep analysis is powered by Philips Wearable Sensing algorithms. This clinical trial is using wrist-worn PPG to analyze heart rate variability and an accelerometer to measure body movements, sleep stages and sleep statistics were automatically computed from overnight recordings. Sleep–wake, 4-class (wake/N1 + N2/N3/REM) and 3-class (wake/NREM/REM) classifiers were trained on 135 simultaneously recorded PSG and PPG recordings of 101 healthy participants and validated on 80 recordings of 51 healthy middle-aged adults. The sleep–wake classifier obtained an epoch-by-epoch Cohen’s κ between PPG and PSG sleep stages of 0.55 ± 0.14, sensitivity to wake of 58.2 ± 17.3%, and accuracy of 91.5 ± 5.1%. κ and sensitivity were significantly higher than with actigraphy (0.40 ± 0.15 and 45.5 ± 19.3%, respectively).
The Effectiveness of Utilizing HRV Indices as a Predictor of ACFT Performance Outcomes
The Army Combat Fitness Test (ACFT) is a performance assessment used by the U.S. Army to assess a cadet’s strength, endurance, and agility with a series of six events to ensure that cadets are combat ready. Heart rate variability (HRV) is an instrument that measures cardiac autonomic modulation. SI was measured and calculated by using the CardioMood (sister company Corsano) equation n(SI = [Amplitude of Mode%]/[2*Mode* (RRmax – RRmin)]
Military Medicine, usad009, https://doi.org/10.1093/milmed/usad009
Please contact us for the full overview of all clinical trials. Please contact us to discuss how we can help you with Raw IP-Free Data, providing an open-source, rich dataset that can be used to develop new diagnostic tools, study disease progression, and innovate personalized healthcare solutions.