Clinical Trials
Clinical Trials
Corsano Cardiowatch Bracelets enable continuous monitoring with multiple algorithms. Corsano is working closely with cardiologists, scientists, hospitals, patients, and research organisations. Scientific research demonstrates the legitimacy of Cardiowatch 287 algorithms.
The Corsano Cardiowatch 287 is enrolled in following selected Clinical Trials:
Photoplethysmography to detect circulatory arrest: A study in patients with induced cardiac arrests
Title
Photoplethysmography to detect circulatory arrest: A study in patients with induced cardiac arrests
Rationale
Out-of-hospital cardiac arrest (OHCA) is a leading cause of global mortality. Delays to initiation of cardiopulmonary resuscitation (CPR) should be as short as possible to achieve the highest survival chance. To reduce treatment delays automated cardiac arrest detection and call for help would be ideal. Photoplethysmography (PPG) is an optical technique often used in sport/smartwatches to monitor heart rhythm at the skin and based on previous studies it seems to have potential to detect circulatory arrest.
Objective
To study photoplethysmography signals during 1) induced circulatory arrest; 2) spontaneous circulatory arrest; 3) cardiac rhythms without circulatory arrest, and to construct a PPG-based algorithm for circulatory arrest detection.
Study design
Prospective multicenter observational study.
Study population
1) Patients undergoing defibrillation testing after (subcutaneous) implantable cardioverter defibrillator (ICD) implantation; 2) Patients undergoing rapid ventricular pacing during transcatheter aortic valve replacement (TAVR); 3) Patients undergoing ventricular tachycardia ablation; 4) Post-cardiac arrest patients.
Intervention
Patients will be equipped with Corsano Cardiowatch 287-2 to record PPG signals during hospital stay including during induced/spontaneous circulatory arrests. The circulatory arrest inductions are part of routine practice during above-described procedures and are not a study-related intervention.
Main study parameters/endpoints
Primary endpoints: Sensitivity and specificity for circulatory arrest. Secondary endpoints: Positive and negative predictive values for circulatory arrest, PPG-signal characteristics.
Nature and extent of the burden and risks associated with participation, benefit and group relatedness
Given the observational nature of this study and the non-invasive comfortable wristband used for PPG signal recording, the medical risk associated with study participation is negligible. There might be potential psychological impact of study participation for post-cardiac arrest patients, which may include settings involving re-arrests or palliative care settings.
Number of Participants
280 patients consisting of an estimated number of n=105 TAVR-patients (event rate 1/1), n=80 ICD-patients (event rate 1/1), n=10 VT-ablation patients (event rate 1/3) and n=60 post-cardiac arrest patients (event rate 1/5). Patients in whom ventricular fibrillation induction or rapid ventricular pacing does not result in complete circulatory arrest (as determined on (invasive) blood pressure measurements) will be replaced by new subjects to guarantee the anticipatednumber of true events.
Inclusion / Exclusion criteria
Inclusion criteria:
- Undergoing ventricular fibrillation induction during defibrillation testing after ICD implantation
- Undergoing rapid ventricular pacing during TAVR procedure
- Undergoing ventricular tachycardia ablation
- Post-cardiac arrest patients
- Additionally, a subject must meet all of the following criteria:- Age => 18 years- Fitting the wristband
Exclusion criteria:
- Unwilling or unable to provide informed consent
- Known hemodynamically relevant subclavian artery stenosis
- Medical issues that interfere with wearing of the wristband (e.g. skin disorders)
- Unavailability of wristband used for PPG recording
Study Centres
RadboudMC, ErasmusMC, LUMC, Isala
Start time, Duration
November 2021. Expected study duration is 2 years
Multi Parameter Vital Signs Monitoring by the Corsano Cardiowatch 287-2 Validation Study
Title
RECAMO: REmote CArdiac MOnitoring by the Corsano Cardiowatch 287-2 of BP and AF
Rationale
Wearables have the potential to monitor patients remotely. The Corsano CardioWatch 287-2 is such a medical device that can monitor atrial fibrillation and long-term blood pressure. The device has been validated using clinical trials in hospitals, but evaluation in the intended remote setting is lacking.
Study design
The study is a single center, single arm prospective study
Primary objective
To compare the number of episodes of atrial fibrillation detected by the Corsano CardioWatch 287-2 during 28 days of use with the number of episodes of atrial fibrillation detected by conventional Holter monitoring during 48 hours of use (standard care).
Secondary objective
To assess the difference in blood pressure measurements obtained by the Corsano CardioWatch 287-2 and the conventional cuff blood pressure monitor over a period of 28 days; to assess the usability of the Corsano CardioWatch 287-2 from a patient perspective.
Study population
One group (A) receiving an EKG monitoring holter for the duration of 24 hours as part of standard care for atrial fibrillation screening. Besides, one group (B) receiving an automatic blood pressure cuff for the duration of 24 hours as part of standard care for blood pressure evaluation.
Main study parameters/endpoints
(1) Absolute percentage increase of patients in whom at least one event of atrial fibrillation is detected by the Corsano CardioWatch 287-2 during 28 days of monitoring, compared to a conventional EKG holter during 24-48 hours of monitoring.
(2) Mean blood pressure difference and its SD between blood pressure measured by the Corsano CardioWatch 287-2 and blood pressure measured by a conventional oscillometric blood pressure cuff.
Secondary study parameters/endpoints
(1) Bias and limits of agreement between blood pressure measured by the Corsano CardioWatch 287-2 and blood pressure measured by a conventional oscillometric blood pressure cuff.
(2) Usability of the Corsano CardioWatch 287-2 in a remote care setting.
Number of Participants
160 patients at the catheterization laboratory of Reinier de Graaf Gasthuis Delft during ABPM or Holter exam, measurements will take place by the Corsano CardioWatch 287-2 and a reference device.
Inclusion / Exclusion criteria
Inclusion criteria:
- ≥ 18 years old;
- undergoing coronary angiography;
- able to provide consent
Exclusion criteria:
- Unable to wear the Corsano CardioWatch 287 due to reasons such as allergic reactions, wounds, amputations etc.;
- Unable to receive blood pressure measurements per cuff due to lymphedema, amputation, dialysis shunt, wounds, etc.;
- Pregnant women;
- Breastfeading women;
Study Centre
Start time, Duration
May 2023, 6 months
Multi Parameter Vital Signs Monitoring by the Corsano Cardiowatch 287-2 Validation Study
Title
MULTI-VITAL Multi Parameter Vital Signs Monitoring by the Corsano Cardiowatch 287-2 Validation Study
Rationale
Today, continuous monitoring of vital signs remains a challenge since it generally requires the patient to be connected to multiple wired sensors, which restricts patient mobility in the intra-mural setting and complicates home monitoring in the extra-mural setting. Wearable devices, although emerging, are often not clinically validated or limited to the monitoring of one or two vital signs.
Objective
This study aims to validate the Corsano CardioWatch 287-2 Bracelet for the continuous monitoring of heart rate at ≤4 bpm root mean squared error (RMSE); interbeat intervals at ≤ 50 ms RMSE; breathing rate at ≤ 2 brpm RMSE; peripheral oxygen saturation at ≤ 3 percentage point RMSE; non-invasive blood pressure at ≤ 10 mmHg RMSE against a reference device.
Study design
The study is a single center, single arm prospective study
Study population
Study participants will be drawn from patients undergoing heart catheterization exam in the Reinier de Graaf Gasthuis
Intervention (if applicable)
The Corsano CardioWatch 287-2 will be put on the patient’s wrist, enabling the measurement of pulse variations. Continuous blood pressure measurements are recorded during the phase that indwelling arterial catheters are present for angiography. Besides, non-invasive blood pressure, peripheral oxygen saturation, heart rate and respiration rate will be measured by a reference device.
Main study parameters/endpoints
Root mean squared error between measurements recorded by Corsano CardioWatch 287-2 and reference device.
Nature and extent of the burden and risks associated with participation, benefit and group relatedness
Patients will be asked for informed consent. If consent is provided, the patient will be put on the Corsano CardioWatch 287-2 right before the heart catherization examination. Besides, the patient will be connected to sensors from a reference monitoring device. When the procedure is finished, the Corsano CardioWatch 287-2 and the sensors will be removed, directly. There will be no follow-up.
Number of Participants
160 patients at the catheterization laboratory of Reinier de Graaf Gasthuis Delft during catheterization exam, measurements will take place by the Corsano CardioWatch 287-2 and a reference device.
Inclusion / Exclusion criteria
Inclusion criteria:
- ≥ 18 years old;
- undergoing coronary angiography;
- able to provide consent
Exclusion criteria:
- who cannot wear the Corsano CardioWatch 287 due to reasons such as allergic reactions, wounds, amputations etc.;
- unable or not willing to sign informed consent;
- with significant mental or cognitive impairment;
- who do not have a suitable entry site for the invasive arterial line.
Study Centre
Start time, Duration
November 2021, 14 months
https://clinicaltrials.gov/ct2/show/NCT05566886
The potential of a wearable monitoring bracelet for COPD patients
Title
Rationale
Chronic obstructive pulmonary disease (COPD) is a chronic inflammatory lung disease characterized by long-term respiratory symptoms and airflow limitation. It affects millions of people worldwide and places a significant economic and health burden on societies and their patients. Exacerbations are an acute and sustained worsening of symptoms, which are associated with significant morbidities such as increased healthcare utilization, poor health status and death. When exacerbations are treated promptly, recovery time and risk for future hospital admissions can be reduced. Hence, there is a need for early detection of COPD exacerbations is evident, as early intervention is a key factor in improving outcomes and preserving quality of life. Emerging technologies in telemedicine and remote patient monitoring can support pro-active management of patients at risk of COPD exacerbations. The Corsano Cardiowatch is a wearable monitoring bracelet that enables continuous monitoring of vital parameters (e.g., heart rate, heart rate variability, breathing rate, atrial fibrillation, physical activity, sleep, temperature), which could be used as a tool to support timely recognition of exacerbations.
Objective
The primary objective of this study is to explore the potential use and value of the Corsano Cardiowatch in monitoring patients with COPD at high risk of an exacerbation, in terms of timely recognition and treatment of an exacerbation. More specifically, we aim to explore its potential 1) during the first 2 months after a hospital admission due to an exacerbation, and 2) in the longer-term. The secondary aim of this study is to investigate potential relevant factors hindering or facilitating the use and implementation of the bracelet as identified by its end-users (i.e., patients and healthcare professionals).
Study design
The study comprises a qualitative design. Two semi-structured focus group interviews will be conducted with relevant stakeholders; one with patients with COPD and one with respiratory healthcare professionals (HCPs). The interviews will be conducted online with 4 to 6 participants and will have a duration of 60 minutes for the HCPs and 75 minutes for the patients respectively.
Study population
We aim to recruit a total of 4 to 6 patients with COPD and a total of 4 to 6 respiratory HCPs (i.e., pulmonologist and nurse specialists). Both patients and HCPs will be recruited in 2 hospitals: Alrijne Hospital Leiderdorp and Franciscus Gasthuis&Vlietland in Rotterdam. Inclusion criteria for patients are 1) being aged 18 years or older, 2) having a diagnosis of COPD, 3) having been admitted to the hospital for an acute exacerbation of COPD in the past 2 years 4) being able to understand, read and speak the Dutch language. The inclusion criteria for HCPs will be that they are treating patients with COPD in one of the participating hospitals.
Main study parameters/endpoints
Expectations, opinions, and attitudes of HCPs and patients regarding the potential of Corsano Cardiowatch for patients with COPD at high risk of exacerbations. More specifically, 1) the potential use and value of the Corsano Cardiowatch during the first 2 months after a hospital admission due to an exacerbation, 2) the potential use and value of the Corsano Cardiowatch in the long-term, 3) potential facilitators and barriers for the use and implementation of the Corsano Cardiowatch.
Number of Participants
12 patients with COPD or respiratory HCPs
Inclusion / Exclusion criteria
Inclusion criteria:
- being aged 18 years or older
- having a diagnosis of COPD
- having been admitted to the hospital for an acute exacerbation of COPD in the past 2 years
- being able to understand, read and speak the Dutch language.
Exclusion criteria:
- N/A
Study Centre
National eHealth Living Lab (NeLL)
Leiden University Medical Center (LUMC)
Start time, Duration
September 2022, 4 months
Phenotyping and Classifying Asthma Exacerbations (ExCluSie-F)
Title
An observational study in patients between 12 and 70 years old with an acute asthma exacerbation, to determine the relation between phenotypical characteristics and the treatment response.
Topic
Asthma is a heterogeneous inflammatory respiratory disease affecting 8 - 9% of the European population. Acute asthma exacerbation (AAE) is characterized as an acute worsening of symptoms and is treated inconsistently with steroids with or without antibiotics. In order to adjust and personalise exacerbation treatment, phenotyping and classifying of asthma exacerbations would be required. Therefore, we want to classify patients with AAEs phenotypically in relation to the treatment response.
Objective
The primary objective of the study is to determine the relationship between exacerbation treatment response at day 7 and the phenotypical characteristics of asthma exacerbations. Secondary objectives are 1) developing a prediction model based on biomarkers and/or clinical data to predict the treatment response of AAEs 2) comparing the environmental, inflammatory, microbiological and lipid parameters of patients diagnosed with asthma between exacerbation phase and recovery (baseline).
Study design
A prospective cohort study in a tertiary asthma centre. Study population: Patients aged 12 - 70 years, diagnosed with mild to severe asthma according to the Global Initiative for Asthma (GINA) guidelines. Patients will be included at the onset of a severe asthma exacerbation.
Devices
Spirometry, Fractional Exhaled Nitric Oxide (FeNO), Corsano Cardiowatch 287-2, e-Nose
Main study parameters/endpoints
Primary endpoint is the relation of phenotypical characteristics with treatment response at day 7, defined by 1) the physician - and patient rated global evaluation of treatment effectiveness (GETE) score 2) difference in Asthma Control Questionnaire 5 (ACQ-5) (> 0.5) 3) difference in handheld spirometry values like forced expiratory volume (FEV1 ≥ 10%). Treatment response will be classified as excellent, good, moderate or poor. Secondary endpoints are 1) a prediction model for the treatment response of AAE 2) aetiology of the AAE 3) blood and local respiratory parameters; microbiota composition; lipid metabolomics and volatile compounds composition at baseline and AAE.
Number of Participants
200 Patients with an asthma exacerbation who are diagnosed with mild to severe asthma according to the GINA guidelines.
Inclusion / Exclusion criteria
Inclusion criteria:
- Patient diagnosed with asthma according to the GINA guidelines between 12 and 70 years old. If patients are doctor's diagnosed with asthma based on clinical data, the further diagnostics will be performed to confirm the asthma diagnosis after the AAE.
- Mild to severe asthma, treated according to GINA guidelines with medium - or high dose inhaled corticosteroids (with or without LABA) or treated with a low dose inhaled corticosteroids combined LABA or leukotriene - receptor antagonist.
- Asthma exacerbation, indicated for systemic corticosteroids.
- Written personal and/or parental informed consent, prior to any study procedures.
- Eligibility and willingness to present during an asthma exacerbation at the Franciscus Gasthuis hospital.
- Ability to use e - health applications.
Exclusion criteria:
- Recent oral corticosteroid treatment in the last 6 weeks.
- Immunosuppressive maintenance medication (azithromycin, systemic corticosteroids maintenance therapy and other) or recently (< 6 weeks) discontinued these medications. (Desensitization therapy indicated for allergies can be included in the study)
- Maintenance medication or recently discontinued (< 6 weeks) biologicals.
- Other underlying inflammatory or auto-immune diseases, such as rheumatologic disease.
- Involvement in the planning and/or conduct of the study (applies to both investigator staff and/or staff at the study site)
- Pregnancy, because of the possible altered immunological status.
- Participation in an interventional study or randomised controlled trial.
Study Centre
Start time, Duration
August 2022, 24 months
Validation Respiration Rate (BRPM) compared with Polygraphy
Title
Validation of Respiratory Rate with the Corsano Cardiowatch 287 Bracelet compared with Polygraphy
Topic
Evaluation of the performance and efficacy of the Corsano Cardiowatch 287 Bracelet in measuring respiratory rate at rest, as compared to golden standard and predicate device.
Number of Participants
25 subjects (at least 30% of each gender)
Inclusion / Exclusion criteria
Inclusion criteria:
People ≥ 18 years old
Exclusion criteria:
Wearer of cardiac implanted electronic device (Pacemaker, ICD)
CardioWatch 287 cannot be worn due to comprehensible reasons (allergic reactions, wounds, amputations, other)
Unable or not willing to sign informed consent
Significant mental or cognitive impairment
Currently enrolled in another clinical investigation in which the intervention might compromise the safety of the subject's participation in this study
Study Design
The patients are selected from the patient population referred for an overnight Polygraphy exam for a suspicion of sleep-disordered breathing. Patients are scheduled for a Respiratory Rate measurement, independent of the proposed study. Patients that are scheduled for these examinations and meet the inclusion criteria (see below) are approached by their practitioner or specialized nurse supervised by the for participation in the trial.
Target Points
The main study endpoints are the Percentage of respiratory rates of matching the resting Polygraphy recording. Percentage of respiration rate at rest ±2 BPM (beats per minute) matching the polygraphy device and manual counting.
Measurement procedures
Participants are requested to wear a smartwatch or bracelet in addition to a Polygraph. Simultaneously, they will have the connected smartphone with them.
Study Centre
Haaglanden Clinics (NL)
Start time, Duration
November 2020, 6 months
Safety and efficacy of anti-CD19 CAR T in subjects with Chronic Lymphocytic Leukemia or Small Lymphocytic Lymphoma
Title
A study evaluating the safety and efficacy of anti-CD19 CAR T in subjects with Chronic Lymphocytic Leukemia or Small Lymphocytic Lymphoma.
Topic
A phase I/II, multicenter study evaluating the feasibility, safety, and efficacy of point-of-care manufactured 19CP02 in subjects with relapsed/refractory B-cell non-Hodgkin lymphoma.
Number of Participants
50 subjects
Study Design
Corsano will deliver CardioWatch 287-2 with CellPoint branded version of Corsano Trials to interact with CellPoint trial patients and to simplify the way patient data is collected and processed (answers to questions, vital signs, etc.). The App will facilitate interaction with patients.
Inclusion Criteria
- Signed informed consent form
- 2. Age ≥ 18 years
- Histologically confirmed diagnosis of CD19+ CLL or SLL with an indication for therapy according to iwCLL criteria, Richter’s transformation is allowed
- Documented relapsed or refractory disease after at least 2 prior lines of therapy:
• Subjects must have been exposed to Bruton tyrosine kinase (BTK)-inhibitors (e.g. ibrutinib, acalabrutinib)
• Richter’s patients who failed a BTK-inhibitor are eligible regardless of number of prior lines of therapy received - Measurable disease according to iwCLL
- ECOG performance status of 0 or 1
- Adequate bone marrow function defined as:
• Absolute neutrophil count (ANC) ≥ 500/μL or ≥ 0.5 × 109/L (without G-CSF support within 7 days of the laboratory test or pegylated G-CSF support within 14 days of the laboratory test)
• Platelet count ≥ 50.000/μL or ≥ 50 x 109/L (without prior platelet transfusion within 7 days before the laboratory test)
• Absolute lymphocyte count ≥ 300/μL or ≥ 0.3 × 109/L;
• CD3+ count ≥ 150/μL or ≥ 0.15 × 109/L - Adequate renal, hepatic and pulmonary function defined as:
• Serum albumin ≥ 3.4 g/dL
• Creatinine clearance (Cockcroft Gault) ≥ 30 mL/min
• Aspartate aminotransferase (AST) ≤ 3 × upper limit of normal (ULN)
• Alanine aminotransferase (ALT) ≤ 3 × ULN
• Total bilirubin ≤ 2 x ULN, except in subjects with Gilbert’s syndrome
• No clinically significant pleural effusion
• Baseline oxygen saturation > 92% on room air - Women of childbearing potential must have a negative serum pregnancy test at screening and prior to the first dose of cyclophosphamide and fludarabine
- Women of childbearing potential and all male subjects must agree to use highly effective methods of contraception (failure rate of < 1% per year when used consistently and correctly) and agree to remain on a highly effective method of contraception from the time of signing the informed consent form until at least 6 months after BCN-CP01 infusion. Subjects must agree to not donate eggs or sperm during this period.
Exclusion Criteria
|
Study Centre
Major Pharma Client (NL)
Start time, Duration
February 2022, 36 months
PPG AF-Burden Clinical Trial CCN-AMC
Title
Determine AF Burden with PPG Trial – Detection and Quantification of Episodes of Atrial Fibrillation Using a Wearable with Photoplethysmographic (PPG) Sensor
Topic
Evaluation of the performance of the Corsano Cardiowatch 287 Bracelet in identifying AF episodes and determining the AF burden in patients with known paroxysmal AF
Number of Participants
100 patients with known paroxysmal atrial fibrillation
Inclusion / Exclusion criteria
Inclusion criteria:
- Known paroxysmal atrial fibrillation
- Patient ≥ 18 years old
- Written informed consent as documented by signature from the patient
Exclusion criteria:
- Cardiac implanted electronic device (Pacemaker, ICD)
- Bracelet cannot be worn due to comprehensible reasons (allergic reactions, wounds, amputations, other)
- Unable or not willing to sign informed consent
- Significant mental or cognitive impairment
- Currently enrolled in another clinical investigation in which the intervention might compromise the safety of the subject's participation in this study
Study Design
In this trial patients with known paroxysmal AF wear a PPG wearable (provided by Corsano) and measure continuous PPG over 48h.The PPG data will be stored on a smartphone app connected to the wearable. Simultaneously the ecg signal will be recorded continuously by holter ecg. The PPG data will be downloaded manually and the AF algorithm analyzes the PPG data to screen for AF episodes. The ecg data will be evaluated by two independent cardiologists blinded to each other and in agreement with the aim to identify AF episodes. The results of the PPG analysis and the results of the ecg evaluation by cardiologists will be compared.
Target points
Primary outcome:
- Detected atrial fibrillation (in comparison to simultaneously recorded continuous Holter-ECG)
Secondary outcomes:
- Overall duration of the atrial fibrillation episodes and AF burden per 24h
- Asymptomatic episodes of atrial fibrillation
Measurement procedures
Reference for assessing the performance and accuracy of the AF algorithm are the diagnoses of the cardiologists based on the ECG data.
Study Centre
Cardiology Centra Netherlands Location UMC
Start time, Duration
Q1 2021, 12 months
Accuracy of CardioWatch 287-2 during profound hypoxia CMC
Title
Accuracy of CardioWatch 287-2 with profound hypoxia (CIV-21-12-038447)
Topic
The present study is looking for calibration and accuracy of SpO2, Pulse Rate and Respiration Rate derived by a newly developed based wrist device (CardioWatch 287-2 or Test Device). These parameters will be computed from raw PPG data delivered by Test Device based on values of a benchmark comparator:
- A well-known benchmark SpO2 sensor of Nellcor (PM10N), including its calculated pulse rate: The Non-Invasive study (NI study) branch.
- Oxygen saturation in arterial blood samples measured by co-oximetry and Nellcor SpO2 will be used to define the accuracy of the test device: The Invasive study (IN study).
- Respiratory rate is provided by measuring end tidal CO2 peaks by a capnometry in both studies.
Individuals undergo progressive hypoxia (10 min / % oxygen decrease) to 10 % of ambient O2 in a normobaric hypoxia room resulting in a subject’s SpO2 or SaO2 range between 97 to 100% and 73%. Nellcor’s finger clip, the wrist CardioWatch 287-2 and a small plastic tube placed near the mouth are attached to the volunteers before and during hypoxia.
Number of Participants
In the first NI study, 12 to 20 healthy volunteers are included, because some minor refinement of the software in the Test Device to improve PPG signals is allowed in the first volunteers. This NI study stops after completing 12 volunteers with the final software version of the Test Device.
In the second IN study, released after completion of the NI study, an arterial catheter is placed to 12 volunteers to obtain arterial oxygen saturation measured by co-oximetry at several time points during progressive hypoxia (25 blood samples/person). Thereafter the accuracy (Arms) of the test device’s SpO2 will be computed.
Per FDA guidance, at least 2, or 15% of the subjects will have dark skin. Equal numbers of men and women will be enrolled.
Inclusion / Exclusion criteria
Inclusion Criteria:
- The subject is male or female, aged ≥18 and <65.
- The subject is in good general health with no evidence of any medical problems.
- The subject has provided informed consent and is willing to comply with the study procedures.
Exclusion criteria:
- Heavy smokers or individuals exposed to high levels of carbon monoxide that result in elevated carboxyhemoglobin levels.
- Individuals subject to conditions that result in elevated levels of methemoglobin
- Individuals with hypoxia (SpO2 < 95 % at 21 % of oxygen)
- Severe claustrophobia
- Subject is known with altitude disease
- The subject is obese (BMI ≥ 31 kg/m2).
- The subject has a known history of moderate to severe heart disease, lung disease, kidney or liver disease.
- The subject is diagnosed with moderate to severe asthma.
- The subject is known with a hemoglobinopathy or history of anemia, e.g. sickle cell anemia, thalassemia, ... who, in the opinion of the investigator, would make them unsuitable for study participation.
- The subject has any other serious systemic illness.
- Any injury, deformity, or abnormality at the sensor sites that in the opinion of the investigators would interfere with the sensors working correctly.
- The subject has a history of fainting or vasovagal response.
- The subject has a history of sensitivity or allergy to local anesthetics or disinfectants.
- The subject has a diagnosis of Raynaud’s disease.
- The subject has unacceptable collateral circulation based on exam by the investigator.
- The subject is pregnant, lactating or trying to get pregnant.
- The subject is unable or unwilling to provide informed consent or is unable or unwilling to comply with study procedures.
- The subject has any other condition, which in the opinion of the investigators would make the subject unsuitable for the study.
- The subject refuses to remove nail-vanish.
Study Design
CardioWatch 287-2 is attached to the wrist. Subjects are in a comfortable semi-recumbent position. Subjects then breathe air mixtures containing reduced amounts of oxygen to produce the desired level of hypoxemia. Stable, safe and controlled hypoxia is achieved breath-by breath by breath respiratory gas analysis and a computer program that permits the inspired gas mixture to be adjusted to achieve a level of lung alveolar gas that will achieve the desired degree of saturation. Typically, saturation levels involve one period with air breathing and then at one of 6 levels with reduced oxygen, e.g. 94%, 90%, 85%, 80%, 75% and 70% saturation. Each level of saturation is held for 30-60 seconds. The operator then changes the inspired oxygen concentration to attain the next desired stead-state level of hypoxia. A "run" consists of several stable steady-state hypoxia levels and together takes 10-15 minutes. Each run is terminated by a breath of 100% O2 followed by room air. 2 Reference oximeters are used, a Nellcor and a Masimo to determine the blood oxygen saturation levels of the subject. The precise target levels of saturation can be adjusted to suit the sponsor, but typical testing is done to satisfy ISO and FDA standards for testing, which is 70% to 100%.
Measurement procedures
The number of subjects and the number of comparisons (paired pulse oximeter readings and arterial saturation values) is determined by current CE-MDR and FDA guidance requirements. This is a minimum of 200 data points and 10 subjects. In the course of this type of study, some subjects may drop out, some readings can be lost due to motion or other interference and occasionally some do not consent.
The following demographic data will be collected on the subjects:
- gender (male, female, other)
- age
- skin tone (Fitspatrick’s skin color scale)
- height (cm)
- weight (kg)
- wrist circumference (cm)
- dominant hand (left or right)
Study Centre
CMC bvba
Europalaan 42b1
3600 Genk
BE 0449.103.169
Principle Investigator
Raf De Jongh, MD, PhD
Start time, Duration
April 2022, Two months
https://clinicaltrials.gov/ct2/show/NCT05542732
Population-based preconception intervention study for a healthy start of life
Generation R Next Study
Population-based preconception intervention study for a healthy start of life
Rationale: The first 1000 days of life reflect the period from the 3 months before conception until the age of 2 years. An accumulating body of evidence suggest that social-demographic background, lifestyle and nutrition of both parents during the preconception and early-pregnancy phases are critical for childhood outcomes. Adverse exposures for the embryo and fetus may lead to developmental adaptations of various organ systems, which are beneficial for short term survival but may lead to adverse health outcomes in later life.
Objective: The Generation R Next intervention study focuses on (future) parents who wish to have or are expecting a child. From previous research within the Generation R cohort, new insights have been developed about the health of (future) parents, reproduction and its effect on the growth and development of the child. This intervention study therefore focuses on improving the health of (expectant) parents by optimizing lifestyle in the preconception period and early pregnancy to improve birth outcomes and long-term outcomes in mother and child.
Study design: Multidisciplinary randomized controlled trial in Rotterdam. The current protocol focuses on the preconception period, the prenatal period and the child age up to 54 months.
Study population: Women and partners living in Rotterdam with a desire to have children or early pregnancy are asked to participate in the study during the preconception period or in the first trimester (gestational age < 11 weeks), respectively. They will be asked to wear CardioWatch 287-1. Recruitment will be through offline and online advertisements and involved caregivers. Then women can register online directly with Generation R Next. Inclusion of participants will take place from September 2022 to February 2024. The aim is to reach a total number of 1750 participating women and 1100 partners (65%).
Intervention: Prior to and/or during early pregnancy, investigations will be performed and an intervention will be offered. The intervention group will attend 3 group sessions (interval 4 weeks) focusing on current national advice for preconception and early pregnancy, lifestyle, coping with stress and additional advice regarding diet. Compliance is encouraged through a digital platform. The intervention and control group will receive a one-off individual lifestyle consultation in line with the current national advice for preconception and early pregnancy (standard care).
Main parameters/endpoints: This study will primarily focus on the following birth outcomes: preterm birth, birth weight (high/low) and low Apgar score at 5 minutes. Secondary outcomes are: (adherence to) lifestyle advice, sleep/wake rhythm, partner support, time to pregnancy, miscarriage, weight gain, stress, hypertension and diabetes during pregnancy, breastfeeding and weight maintenance after pregnancy.
Number of Participants: 1750 participating women and 1100 partners (65%)
Study Centre: ErasmusMC, Rotterdam
Start time, Duration: Expected start date winter 2023, 2 years are needed for inclusion.
Validation PPG measured HR and RR compared with ECG
Title
Validation of Photoplethysmography Measured Heart Rate and RR-intervals with the Corsano CardioWatch 287 Module Compared with ECG
Topic
To study the feasibility, efficacy and performance of the Corsano CardioWatch 287 module with a photoplethysmographic sensor for measuring heart rate and RR-intervals
Number of Participants
100 presumably healthy people
Inclusion / Exclusion criteria
Inclusion criteria:
- presumably healthy people
- People ≥ 18 years old
- non clinical environment
Exclusion criteria:
- Bracelet cannot be worn due to comprehensible reasons (allergic reactions, wounds, amputations, other)
Study Design
Patients fulfilling the inclusion criteria will receive a wearable device (Corsano CardioWatch 287 provided by MMT) and a smartphone with the MMT-App. The smartwatch and the app on the smartphone are coupled. Continuous monitoring of heart rate and –rhythm will be conducted using photoplethysmography (PPG). Simultaneously, a 5 min resting ECG recording will be conducted. Data obtained with the Corsano CardioWatch 287 will be saved on the smartphone and will be downloaded by the physician at the end of the monitoring procedure. Data will then be analysed by trained analysts and supervising physicians at Cardiologie Centra Nederland. The PPG data from the Corsano CardioWatch 287 will be compared to a 5 minute, 12-lead resting ECG recording on the measurement of heart rate and RR-intervals.
Target Points
Main study parameters/endpoints: Percentage of heart rate and RR-intervals of the PPG sensor matching the resting ECG recording
Measurement procedures
Participants are requested to wear a smartwatch or bracelet for 5 minutes in addition to a resting ECG. Simultaneously, they will have the connected smartphone with them.
Study Centre
Cardiologie Centra Nederland, Amsterdam UMC, Location AMC (NL)
Start time, Duration
September 2020, 4 months
Tele-TOKS: Implementation of an early warning system
Title
Tele-TOKS Implementation of an early warning system on a cardiology ward utilizing a wrist-worn medical device, with comparison to conventional measurements taken by nurses
Topic
Vital sign changes often precede cardiac arrest, unexpected death, or ICU admission. If observed and treated, it could prevent the patient's condition from deteriorating. Systematically detecting abnormal vital signs can predict which patients are at risk of death and improve outcomes through timely interventions. TOKS is a Danish algorithm derived from the UK's National Early Warning Score (NEWS). TOKS 2.1 was inspired by New Zealand's Wellington Early Warning Score, which measures vital parameters and calculates an overall score. Cardiowatch 287-1 is a wrist-worn, wireless remote monitoring device for residential and healthcare environments and is CE-certified and EU-MDR-compliant. The device continuously monitors respiratory rate, oxygen saturation, blood pressure, pulse rate, and core temperature from the wrist, as required for automated TOKS 2.1 measurements.
Number of Participants
The study will include at least 100 patients over the age of 18 who are referred to our hospital's cardiology ward.
Target points
Sensitivity and specificity of the Corsano Health Cardiowatch 287-2B wristband in comparison to manual measurements performed by trained nurses.
Study Centre
Regionshospital Nordjylland
Bispensgade 37
9800 Hjørring, Denmark
Start time, Duration
Q4 2022, 6 months
Smart in OAC – AFNET 9
Title
Case-finding Study to Detect and Quantify Episodes of Absolute Arrhythmia Using an Automated, Wearable Monitoring System (Smart in OAC)
Topic
To demonstrate that patients with previously unknown atrial arrhythmias can be detected by a wearable in combination with Preventicus Heartbeats Core using a completely digital patient data capture system.
Number of Participants
Patients to be screened: 1,000
Inclusion criteria
• Informed consent
• Mobile phone compatible with the PPG-wearable (see Appendix 3)
• Stable internet connection
Exclusion criteria
• Unwillingness to participate
• Inability to consent
• Known AF
• Known OAC treatment
Study Design
In this study, the feasibility of evaluating the efficacy of a cloud based analytic service in combination with a Corsano CardioWatch 287 in detecting AA will be assessed and the number of cases found. The design aims to provide simple, low-threshold access to this screening technology targeting at-risk populations. This study will not cause any restrictions on the usual care of the study participants. Access to the screening will be provided close to home and free of charge. The app will also be used to validate and enhance the clinical information about the participants captured during the study. This information will be used to define and refine target groups with highest screening efficiency and, in the long-term, outcome benefits. The study will describe the prevalence of AA in an unselected population that can be reached by a low-threshold screening procedure. The study will also generate important information on the different possible screening environments in different countries (e.g. pharmacies, GP practices, etc). Structures of work-up and continuous patient management in screen-positive individuals will be described and may help to design screening pathways in the main trial. By verifying the wearable-diagnoses by ECG in all screen-positive and a random selection of screen-negative participants, the diagnostic accuracy of the wearable in combination of cloud based analytic service can be estimated. Cost effectiveness assessment will evaluate the cost of low-threshold remote screening per patient identified and help guide to target high risk groups with optimal screening yield in the future. The collected data will provide the sound basis for the design and conduct of a large outcome trial.
Target points
The primary outcome will be
- Number of participants reached
- Number of participants with detected absolute (atrial) arrhythmia.
The secondary outcomes include:
- Rate of participants with newly detected absolute (atrial) arrhythmia
- Rate of participants with confirmed absolute (atrial) arrhythmia (sub-analysis: atrial fibrillation)
Measurement procedures
Reference to evaluate the performance and accuracy of the Preventicus Heartbeats algorithm are the diagnosis of the 14d Holter ecg, analysed by cardiologists.
Study Centre
Germany: Hamburg
UK: Birmingham
Spain: Barcelona
Poland: Krakow (or Katowice)
Start time, Duration
Start: Q1 2021, Total study duration: 12 months
Duration per patient: 2 months
https://clinicaltrials.gov/ct2/show/NCT04579159
Atrial Fibrillation detection in OBESity using Ehealth
Title
Atrial Fibrillation detection in OBESity using Ehealth
Topic
Obesity is a global epidemic. Obesity is associated with an increased risk of atrial fibrillation (AF). AF is the most common sustained cardiac rhythm disorder in humans with potentially life threatening complications. Detection of subclinical AF in obesity patients would allow initiation of proper therapy and follow-up. However, to financially and logistically permit screening of subjects on AF, subjects with the highest risk of having AF should be identified. Currently, knowledge on the mechanisms through which obesity increases the risk of AF remains largely unclear and insufficient to develop such strategies. Franciscus Gasthuis & Vlietland is one of the largest bariatric surgery centers of The Netherlands, with multiple research projects focusing on the obesity patient.
Number of Participants
200 consecutive adult obesity patients undergoing bariatric surgery
Inclusion / Exclusion criteria
Inclusion criteria
- BMI of ≥ 35 kg/m2
- scheduled for bariatric surgery
- age ≥ 50 years
- written informed consent
Exclusion criteria
- Known cardiac disease. Whether a patient has known cardiac disease will be determined by assessment of the available patient files and by asking the patient.
Study Design
The value of AF-screening will be studied in an investigator driven, cross sectional, observational cohort study of obesity patients (primary objective). Selected patients are obesity patients who are screened for bariatric surgery, aged 50 years and older, without a history of cardiac disease. Patients will undergo conventional and advanced echocardiography and laboratory tests as well to investigate signs of subclinical cardiac dysfunction that may be related to AF (secondary objective). Also, a prospective follow-up study of obesity patients undergoing bariatric surgery will be performed to gain insight in the pathophysiology of increased risk of AF in obesity (secondary objective).
Target points
The primary outcome will be
- the proportion of obesity patients with AF as detected by 1 week heart rhythm registration with an AF-detection patch (before bariatric surgery)
The secondary outcomes include:
- Change in the proportion of obesity patients with any AF episode as detected by an AFdetection patch between study onset (1 week monitoring before surgery) and one year after surgery (1 week monitoring)
- Change in the proportion of obesity patients with any AF episode as detected by an ILR between study onset (3 months monitoring before surgery) and in the period of 9 to 12 months after surgery (3 months monitoring)
Study Centre
Franciscus Gasthuis, Rotterdam (NL)
Start time, Duration
Start: 1 september 2020, Total study duration: 18 months
SmartAF – Monitoring of Atrial Fibrillation in Hospitalized Patients for Stroke Prevention
Title
SmartAF: Monitoring of Atrial Fibrillation in Hospitalized Patients for Stroke Prevention
Topic
Atrial fibrillation (AF) is the most common cardiac arrhythmia and a major risk factor for cerebrovascular insults. Screening for AF in hospitalized patients is challenging. The classical ECG monitoring requires the patient to wear patch electrodes and cables, which is uncomfortable and may lead to skin irritation and reduced mobility. In this trial the Corsano CardioWatch 287 will be used to conduct continuous heart rate and –rhythm monitoring in hospitalized patients with moderate to high risk for AF. Collected data will then be automatically de-identified and send to Preventicus for analysis. The algorithm is a clinically validated, CE marked and certified medical device (class IIa). If an arrhythmia is detected, the patients will receive a 7d Holter ecg for confirmation.
Number of Participants
Inclusion / Exclusion criteria
Inclusion criteria
- Hospitalized patients on the internal medicine ward of USB
- CHA₂DS₂-VASc Score ≥ 2
- Written informed consent as documented by signature from the patient
Exclusion criteria
- Current or prior diagnosis of AF
- Chronic anticoagulation therapy for other reasons
- Cardiac implanted electronic device (Pacemaker, ICD)
- Smartwatch cannot be worn due to comprehensible reasons (allergic reactions, wounds, amputations, other)
- Unable or not willing to sign informed consent
Study Design
This is an investigator-initiated, prospective, single-center observational trial to evaluate the performance and accuracy of a diagnostic algorithm provided by Preventicus in identifying hospitalized patients with atrial fibrillation. 250 patients hospitalized in the internal medicine department will be enrolled in this study. Patients without prior history of AF and increased CHA₂DS₂-VASc Score (≥ 2) will be approached and will receive a wearable device provided by MMT for PPG-based continuous heart rate and –rhythm monitoring. Data will be transmitted to a local server via the USB ICT-platform, de-identified and forwarded to Preventicus for analysis. Preventicus will analyse the data files and forward it for a second quality test to Telecare Ulm if an arrhythmia is suspected. If the quality check is positive the likelihood of AF is >90%. In this case the report will be send back to the USB and re-identified. The Sponsor-Investigator will be informed and the respective patient will receive a 14-day Holter ECG. If AF is confirmed, subsequent consultation with a cardiologist will be recommended.
Target points
The primary outcome will be
- Detected AF episodes
The secondary outcomes include:
- AF screening durations
- Health economic simulation: costs per identified AF case after AF screening and after AF confirmation
Study Centre
University Hospital Basel
Start time, Duration
Start: september 2021, 1.5 years
MINDfulness in Cardiac Obesity Rehabilitation using E-health (MINDCORE) – pilot study
Topic
The number of cardiac patients with obesity is rising, however their number of referral to cardiac rehabilitation (CR) is not. Patients with obesity report a higher level of anxiety, a greater fear of movement after a cardiac event, a poor relationship with food and frequently a negative body and self-image. These challenges and behavioural patterns could be the obstacle for obese patients in maintaining a healthy lifestyle after completing the regular CR program after a cardiac event. Mindfulness Based Stress Reduction (MBSR) training is a promising treatment that has not been investigated in this population. MBSR is an 8-week evidence-based group program that employs mindfulness meditation, breathing exercises, body awareness, and exploration of patterns of behaviour. In addition, MBSR improves heart rate variability (HRV) and potentially lowers blood pressure. MBSR could support cardiac obese patients in improving their lifestyle decisions.
Number of Participants
The study will include 20 patients with a Body Mass Index (BMI) >30 kg/m2 with coronary artery disease (CAD) or atrial fibrillation (AF) who fulfill the guidelines for participation in CR.
Inclusion / Exclusion criteria
Inclusion criteria:
Obese patients with a BMI of ³ 30 kg/m2 who are referred to CR with documented coronary artery disease (CAD) (myocardial infarction [ST-segment elevation myocardial infarction; non- ST-segment elevation myocardial infarction], unstable angina pectoris, chronic coronary syndrome) or nonvalvular AF and who fulfill the guidelines for CR participation are included. Patients have to sign informed consent.
Exclusion criteria
- Non-Dutch speaking participant.
- Incapable of understanding and utilizing digital communication.
- Severe psychological or cognitive impairment that limit participation in group interventions.
Study Design
The mindfulness CR study is an investigator driven observational, exploratory pilot study.
A total of 20 patients will be prospectively recruited for participation at locations of “Capri Hartrevalidatie” in Rotterdam and The Hague. The MBSR training will be given in a group format andraining will be given online through Zoom once a week for a total of 8 weeks. All patients will receive the Corsano CardioWatch to wear for the duration of 8 weeks during the MBSR training and one month after completing MBSR to track changes in steps per day, heart rate, and heart rate variability (HRV). Patients will receive questionnaires at baseline, after completion of MBSR, and at 1-, 3- and 6-month follow-up.
Target points
The primary outcome will be to determine the feasibility and protocol implementation of a specially designed MBSR training in CR for obese patients.
The secondary outcomes include:
- To explore whether MBSR in patients with obesity referred to CR leads to an improvement in HRV. HRV will be measured with the Corsano CardioWatch.
- To explore the potential effect of MBSR on kinesiophobia in patients with obesity who are referred to CR.
- To explore the possible effect of MBSR on eating behaviour in patients with obesity referred to CR.
Study Centre
Capri Hartrevalidatie
Start time, Duration
Start: 1 May 2022. Total study duration: 18 months
DivAirCity: Personal exposure to air pollution and associated vital signs
- age ≥ 18 years
- written informed consent
- chronic stress disorder
- Pregnancy
- diabetes or orthopedic or cardiovascular diseases
Participants must perform the exercise routine in three different locations: a green-blue nature environment, a busy street (on the sidewalk/pedestrian mall), and a control environment in an office/classroom. Participants will be accompanied by a researcher during data collection. The required exercises will be performed in a standing position (star jumps) without the need for a mat. Participants will be informed of the study design prior to their participation and will have the discretion to decide what type of assistive devices they will need to complete the required tasks (e.g., special clothing, hydration, etc.). Only the data collection devices will be provided by the researchers and monitored by them throughout the duration of the study to ensure that participants are within the safe limits of their own fitness.
- What impact on psychological stress and physiological metrics does nature-based therapy in varying air quality conditions have?
- Are you able to predict the quality of nature-based solutions given environmental indicators provided by mobile and static sensor arrays?
- How do environmental characteristics such as traffic presence and blue-green space affect air quality?
Generation R Next: Longitudinal study that examines the growth, development, and health of children
Determine AF Burden With PPG Trial - Detection and Quantification of Episodes of Atrial Fibrillation
- age ≥ 18 years
- written informed consent
- Cardiac implanted electronic device (Pacemaker, ICD)
- Smartwatch/Bracelet and/or ECG device cannot be worn due to comprehensible reasons (allergic reactions, wounds, amputations, other)
- Patients unable or not willing to sign informed consent Significant mental or cognitive impairment
Corsano EndoWatch: The smart wearable that helps to improve life of patients after treatment of a suprasellar brain tumour
Realtime monitoring of activity and feedback for patients with femoral bone metastases
Interested in our Trial Programme?
We are currently performing pilots with selected clients. Contact us if you want to know more!
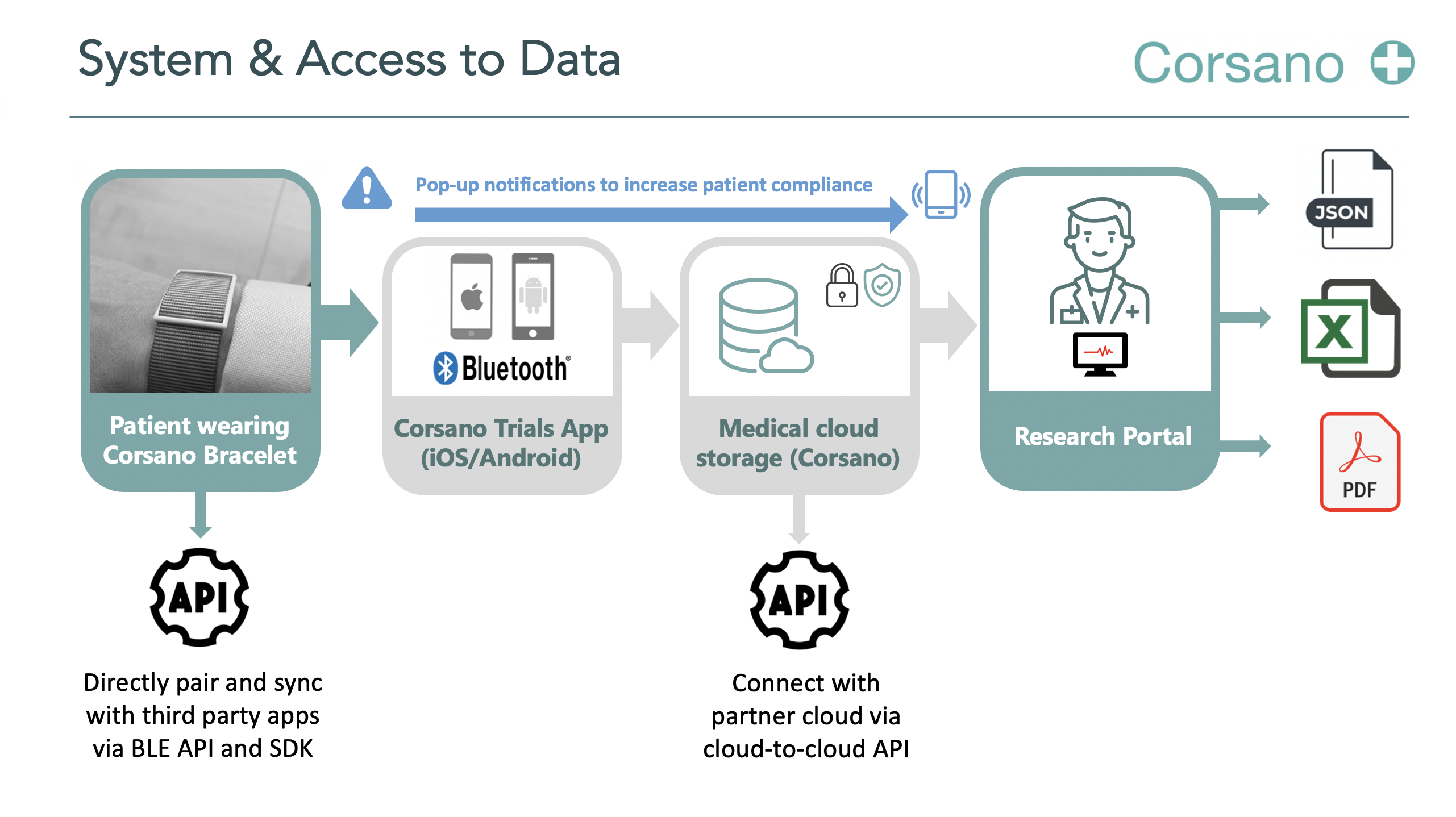
Corsano provides researchers to set which Vital Parameters should be monitored and at which frequency. Corsano offers access to all data, i.e. Raw PPG Green, Red and iR data can be exported up to 128 Hz.
