Title
Accuracy of respiratory rate (RR) measurement by CardioWatch287-2 (CW2) during different respiratory rates (ACW2-RR)
Background
The aim of the present study is to compute the accuracy of CW2 in terms of respiratory rate (RR) in healthy volunteers covering the full breathing rates. Study device is CW2, and benchmark device for RR is physician confirmed respiratory rate defined on basis of the behavior of CO2 measured at the entrance of the nose/mouth of the participants for all volunteers.
Study Design
RR will be obtained by FDA cleared Reference Device GE Medical Monitor B125M
(K213490) with E-MINIC module (K052582). Based on the capnographic CO2, a blinded physician will note/affirm twice a minute the RR value as reference value for computation of CW2’s accuracy.
RR: based on rib cage and abdominal movements RR measured by inductance plethysmography obtained by a FDA cleared Nox-T3 device (K082113). RR reflected by Nox-T3 compared with RR of heathy volunteers was shown to be accurate during the ACW2-MOT study.
12 healthy volunteers, both sexes (at least 30 % of a gender will be represented) and including different Fitzpatrick’s skin types (including type 5 & 6 > 15%) will be asked to breath at different metronomic guided RRs. At least 42 TPIs will show RR in the predefined RR periods and at least 11 TPIs during the different free breathing periods from each volunteer will be used for computing the accuracy of RR reflection by CW2 and RR defined by the physician or NOX-T3 monitor, totaling 53 measuring points per volunteer, > 600 for the total included volunteer population.
Full RR Range 4-60 breaths per minute are to be covered, more specified a RR of 3, 8, 13, 18, 23, 28, 33, 38, 43, 48, 53, 58, 62 will be the predefined. As such this study is interventional.
This study will be executed at CMC, a non-hospital location with altitude room (which will not be used for the present study) in the city of Genk under supervision of Dr. N. De Neve and Dr. R. De Jongh
Study Population
For this interventional study, healthy volunteers will be recruited at Complementair Medisch Centrum (CMC) at Genk. This centre is an altitude centre but during the present study all volunteers stay in normoxia. Only one volunteer per session will be followed by a non-medical and a medical supervisor.
Twelve healthy volunteers (minimum 30% of each gender, minimum 15% with Fritzpatrick’s skin scale of V or VI) will undergo a hypo/hyperventilation protocol to obtain data for the full range 4- 60 bpm in intervals of +/- 5 bpm.
Volunteers will be seated in an arm chair, and an oronasal tube will be placed at the entrance of the mouth/nose for CO2 aspiration. CO2 will be reflected by monitor GE Medical Monitor B125M (K213490) with E-MINIC module (K052582) and this monitor will be used for physician objectivated/noted RR.
Volunteers will be asked to breathe in accordance with a metronome for non-habitual breathing. Before the study, volunteers learn breathing in low tidal volumes to avoid hypocapnia. If necessary, when side-effects of hypocapnia are encountered, periods of free breathing will be prolonged.
Study center
Complementair Medisch Centrum in Genk, Belgium
Start time, Duration
January 2024, 2 months
Interested in our Trial Programme?
Corsano Cardiowatch Bracelets enable continuous monitoring with multiple algorithms. Corsano is working closely with cardiologists, scientists, hospitals, patients, and research organisations. Scientific research demonstrates the legitimacy of Cardiowatch 287 algorithms.
We are currently performing pilots with selected clients. Contact us if you want to know more!
Corsano CardioWatch is a wrist worn medical grade multi sensor bracelet to capture up to 19 vital parameters. Primary use cases are 24/7 continuous clinical trials and remote patient monitoring.
The bracelet monitor offers flexibility to set data collection intervals by minute, by second or 25Hz, 32 Hz or 128 Hz. We measure Heart Rate, RR intervals, Breathing Rate, A-Fib detection, Skin Temperature, Activity and Sleep. SpO2, Core Body Temperature, ECG and Blood Pressure are in development.
Clinical accuracy is paramount for us. In addition to third parties' algorithms, i.e. from Philips, we run our proprietary algorithms to realize superior data accuracy. Corsano technology has been clinically validated in major academic hospitals. The bracelet is manufactured under ISO 13485 and CE-MDR compliance. The process for FDA Clearance has started.
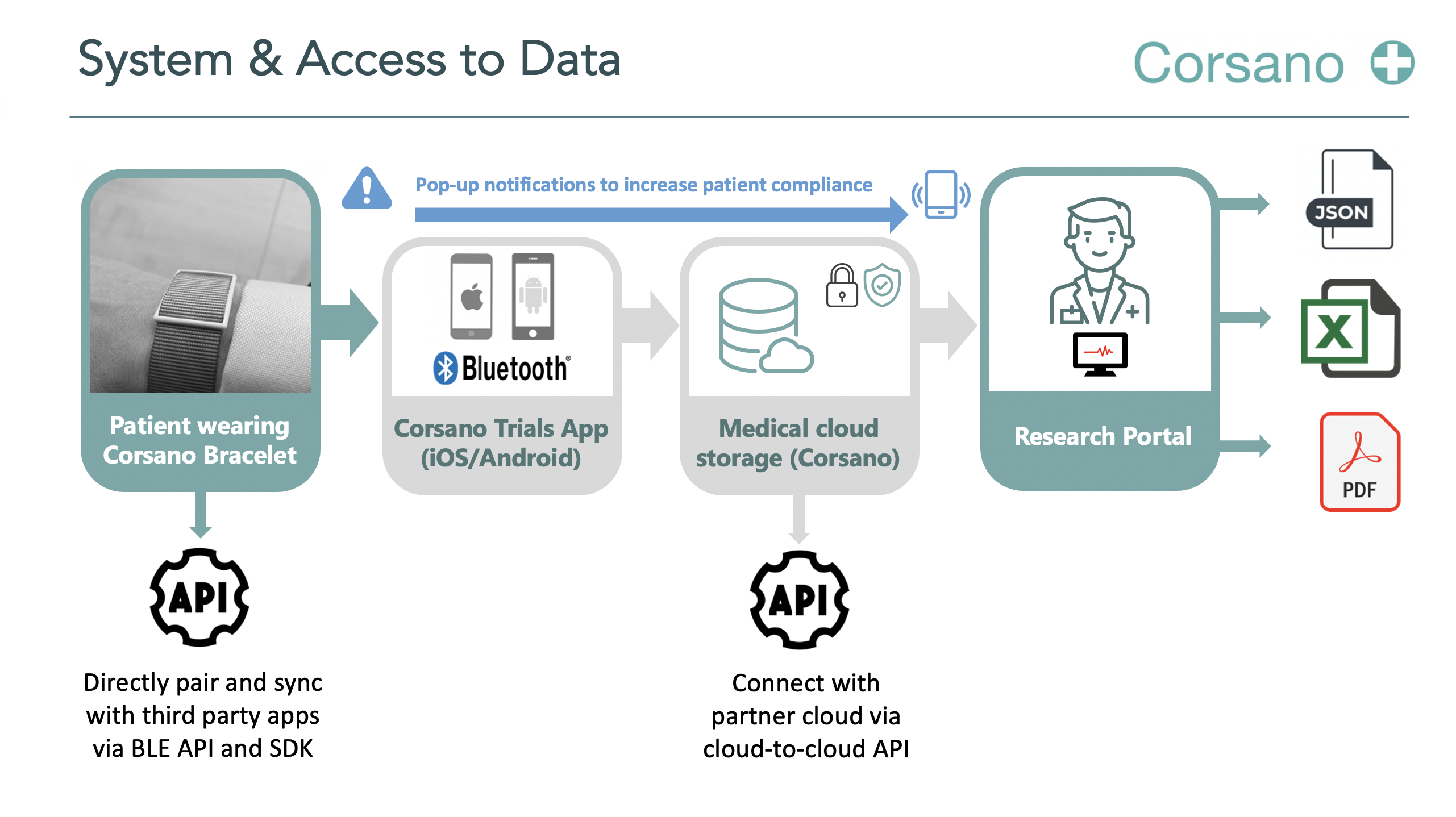
Our end-to-end solution consists of light-weight and comfortable bracelets with a patient research portal. We have developed and produced over 200’000 devices. The Corsano platform has been optimized on connections, data buffers and battery life up to 15 days. We offer access to raw continuous high frequency data through API and cloud-to-cloud solutions.
The founders of Corsano Health have over 100 years of experience in the Swiss Watch industry, with deep experience about ergonomic design and materials for wearables that are worn 24/7.