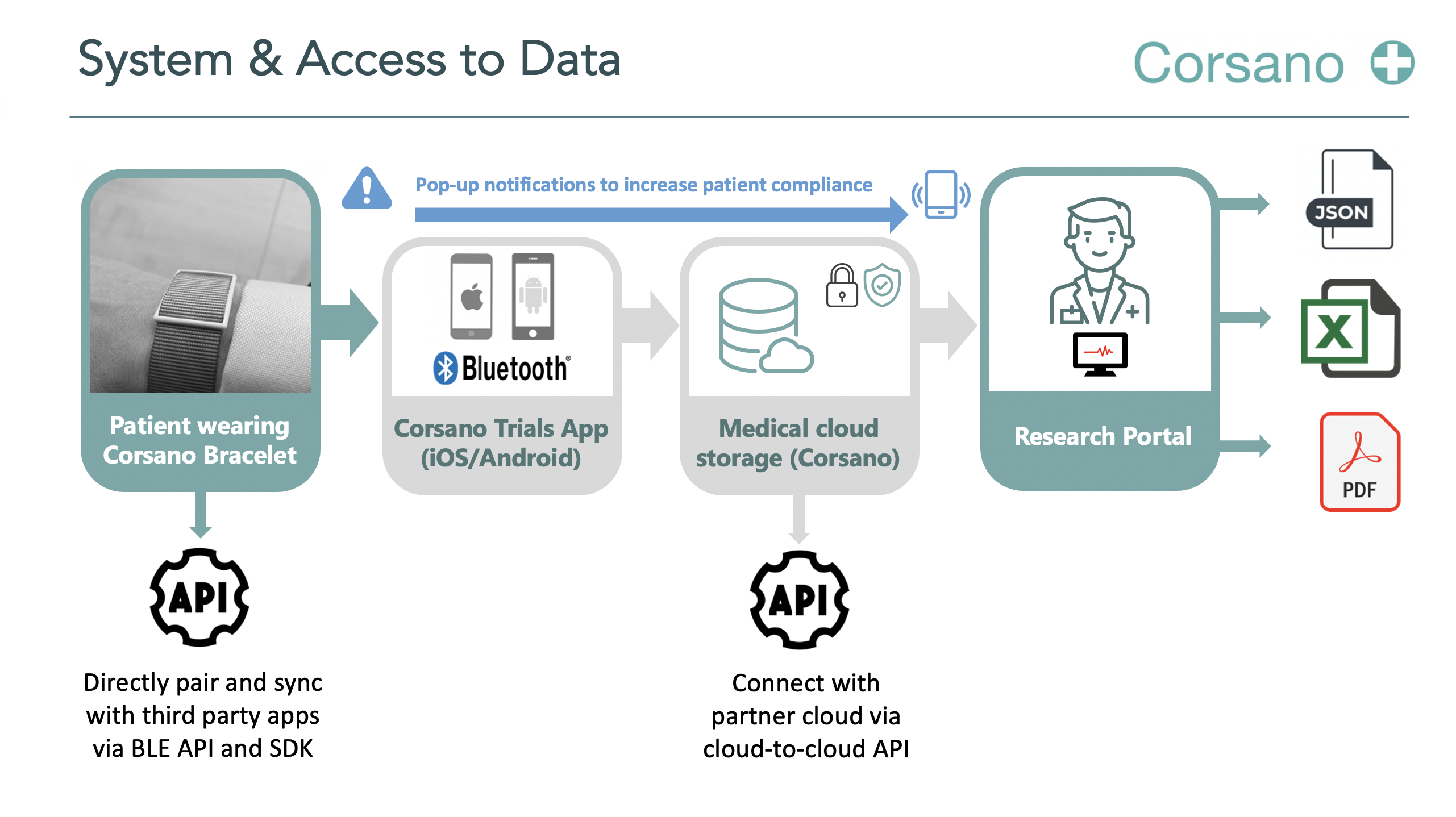
Topic
Bone metastases represent a common complication in cancer patients, particularly in advanced stages of the disease. The presence of bone metastases can cause skeletal-related events, such as bone pain, pathological fractures, spinal cord compression, and hypercalcemia. Therefore, it is essential to monitor bone strength to evaluate the risk of fracture and optimize treatment strategies. Realtime monitoring of activity and feedback to patient what activity they can do will reduce risk of fracture.
Study Design
The BOS (Bone Quality Score) is a numerical score developed by researchers from the University of Twente and the Netherlands Cancer Institute (NKI) to evaluate bone quality in patients with bone metastases. The score was developed by Esther Tank and Nico Verdonschot. The BOS score takes into account several factors that affect bone strength, including bone mineral density, cortical thickness, and the presence of lytic or sclerotic lesions. The score ranges from 0 to 10, with higher scores indicating better bone quality. The BOS score has been shown to be a reliable and valid tool for evaluating bone quality in patients with bone metastases. It can be used to guide treatment decisions and predict the risk of fracture. Follow-up study with Corsano cardioWatch 287-2 will determine how much a patient can load their femur. Aim is to measure activity/functional capacity of patients and what exercises are safe to do for patients with femoral bone metastases.
Study Centre
University of Twente and the Netherlands Cancer Institute, The Netherlands
Start time, Duration
Start: Q4 2023
Interested in our Trial Programme?
Corsano Cardiowatch Bracelets enable continuous monitoring with multiple algorithms. Corsano is working closely with cardiologists, scientists, hospitals, patients, and research organisations. Scientific research demonstrates the legitimacy of Cardiowatch 287 algorithms.
We are currently performing pilots with selected clients. Contact us if you want to know more!
The founders of Corsano Health have over 100 years of experience in the Swiss Watch industry, with deep experience about ergonomic design and materials for wearables that are worn 24/7.