Topic
The number of cardiac patients with obesity is rising, however their number of referral to cardiac rehabilitation (CR) is not. Patients with obesity report a higher level of anxiety, a greater fear of movement after a cardiac event, a poor relationship with food and frequently a negative body and self-image. These challenges and behavioural patterns could be the obstacle for obese patients in maintaining a healthy lifestyle after completing the regular CR program after a cardiac event. Mindfulness Based Stress Reduction (MBSR) training is a promising treatment that has not been investigated in this population. MBSR is an 8-week evidence-based group program that employs mindfulness meditation, breathing exercises, body awareness, and exploration of patterns of behaviour. In addition, MBSR improves heart rate variability (HRV) and potentially lowers blood pressure. MBSR could support cardiac obese patients in improving their lifestyle decisions.
Number of Participants
The study will include 20 patients with a Body Mass Index (BMI) >30 kg/m2 with coronary artery disease (CAD) or atrial fibrillation (AF) who fulfill the guidelines for participation in CR.
Inclusion / Exclusion criteria
Inclusion criteria:
Obese patients with a BMI of ³ 30 kg/m2 who are referred to CR with documented coronary artery disease (CAD) (myocardial infarction [ST-segment elevation myocardial infarction; non- ST-segment elevation myocardial infarction], unstable angina pectoris, chronic coronary syndrome) or nonvalvular AF and who fulfill the guidelines for CR participation are included. Patients have to sign informed consent.
Exclusion criteria
Non-Dutch speaking participant.
Incapable of understanding and utilizing digital communication.
Severe psychological or cognitive impairment that limit participation in group interventions.
Study Design
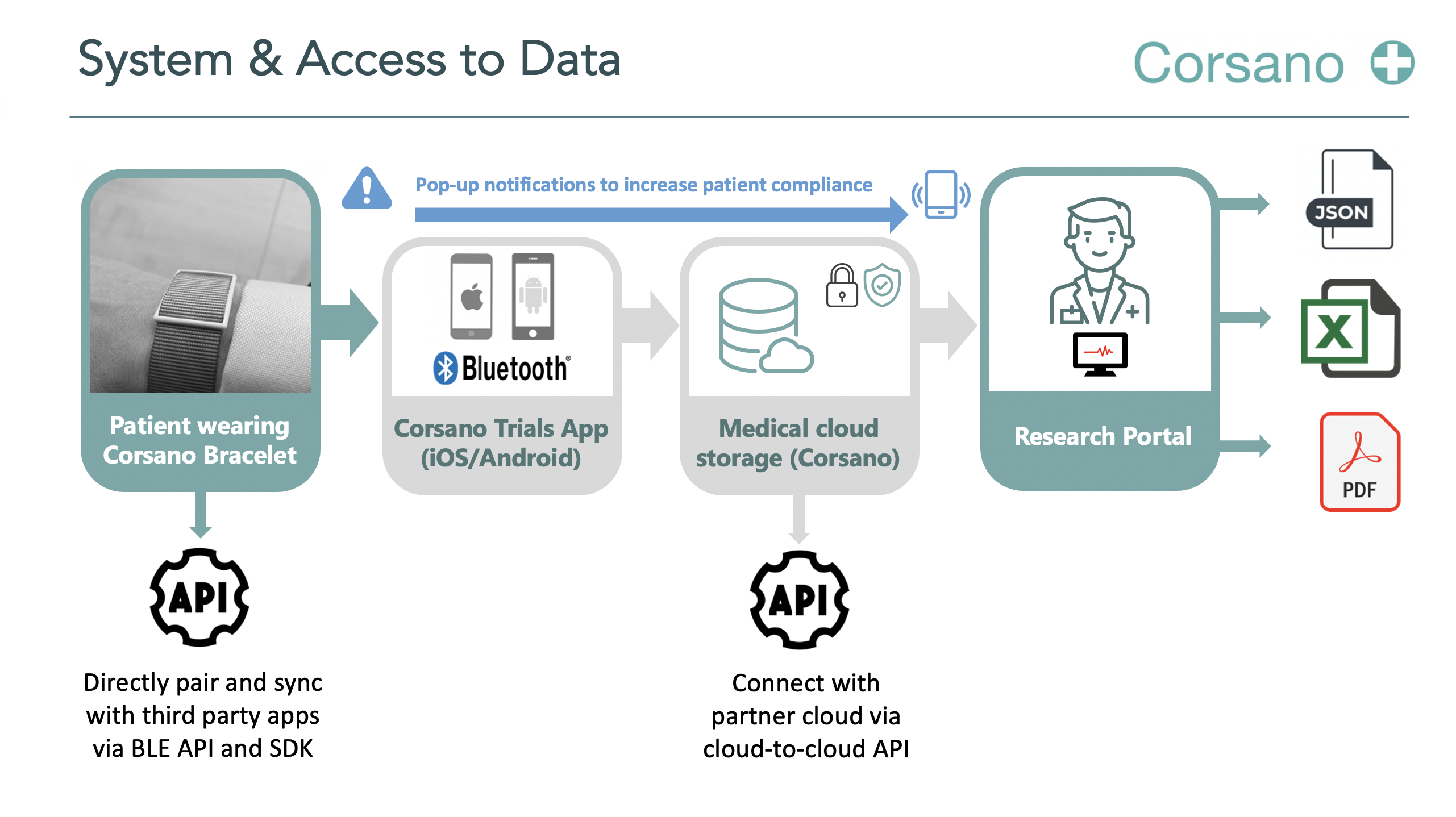
The mindfulness CR study is an investigator driven observational, exploratory pilot study.A total of 20 patients will be prospectively recruited for participation at locations of “Capri Hartrevalidatie” in Rotterdam and The Hague. The MBSR training will be given in a group format andraining will be given online through Zoom once a week for a total of 8 weeks. All patients will receive the Corsano CardioWatch to wear for the duration of 8 weeks during the MBSR training and one month after completing MBSR to track changes in steps per day, heart rate, and heart rate variability (HRV). Patients will receive questionnaires at baseline, after completion of MBSR, and at 1-, 3- and 6-month follow-up.
Target points
The primary outcome will be to determine the feasibility and protocol implementation of a specially designed MBSR training in CR for obese patients.The secondary outcomes include:
To explore whether MBSR in patients with obesity referred to CR leads to an improvement in HRV. HRV will be measured with the Corsano CardioWatch.
To explore the potential effect of MBSR on kinesiophobia in patients with obesity who are referred to CR.
To explore the possible effect of MBSR on eating behaviour in patients with obesity referred to CR.
Study Centre
Capri Hartrevalidatie
Start time, Duration
Start: 1 May 2022. Total study duration: 18 months
Interested in our Trial Programme?
Corsano Cardiowatch Bracelets enable continuous monitoring with multiple algorithms. Corsano is working closely with cardiologists, scientists, hospitals, patients, and research organisations. Scientific research demonstrates the legitimacy of Cardiowatch 287 algorithms.
We are currently performing pilots with selected clients. Contact us if you want to know more!
The founders of Corsano Health have over 100 years of experience in the Swiss Watch industry, with deep experience about ergonomic design and materials for wearables that are worn 24/7.