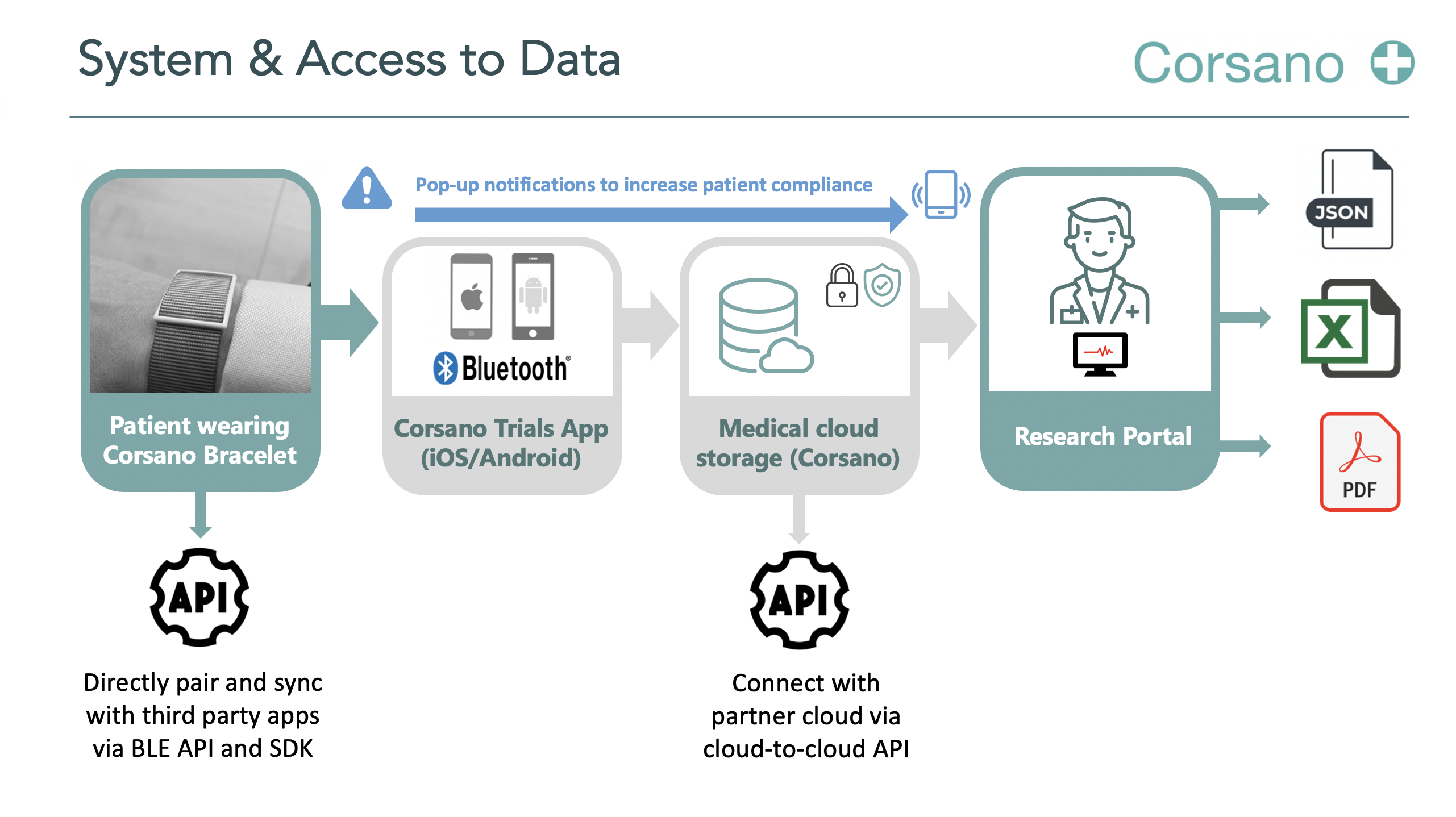
Generation R Next Study
Population-based preconception intervention study for a healthy start of life
Rationale
The first 1000 days of life reflect the period from the 3 months before conception until the age of 2 years. An accumulating body of evidence suggest that social-demographic background, lifestyle and nutrition of both parents during the preconception and early-pregnancy phases are critical for childhood outcomes. Adverse exposures for the embryo and fetus may lead to developmental adaptations of various organ systems, which are beneficial for short term survival but may lead to adverse health outcomes in later life.
Objective
The Generation R Next intervention study focuses on (future) parents who wish to have or are expecting a child. From previous research within the Generation R cohort, new insights have been developed about the health of (future) parents, reproduction and its effect on the growth and development of the child. This intervention study therefore focuses on improving the health of (expectant) parents by optimizing lifestyle in the preconception period and early pregnancy to improve birth outcomes and long-term outcomes in mother and child.
Study design
Multidisciplinary randomized controlled trial in Rotterdam. The current protocol focuses on the preconception period, the prenatal period and the child age up to 54 months.
Study population
Women and partners living in Rotterdam with a desire to have children or early pregnancy are asked to participate in the study during the preconception period or in the first trimester (gestational age < 11 weeks), respectively. They will be asked to wear CardioWatch 287-1. Recruitment will be through offline and online advertisements and involved caregivers. Then women can register online directly with Generation R Next. Inclusion of participants will take place from September 2022 to February 2024. The aim is to reach a total number of 1750 participating women and 1100 partners (65%).
Intervention
Prior to and/or during early pregnancy, investigations will be performed and an intervention will be offered. The intervention group will attend 3 group sessions (interval 4 weeks) focusing on current national advice for preconception and early pregnancy, lifestyle, coping with stress and additional advice regarding diet. Compliance is encouraged through a digital platform. The intervention and control group will receive a one-off individual lifestyle consultation in line with the current national advice for preconception and early pregnancy (standard care).
Main parameters/endpoints
This study will primarily focus on the following birth outcomes: preterm birth, birth weight (high/low) and low Apgar score at 5 minutes. Secondary outcomes are: (adherence to) lifestyle advice, sleep/wake rhythm, partner support, time to pregnancy, miscarriage, weight gain, stress, hypertension and diabetes during pregnancy, breastfeeding and weight maintenance after pregnancy.
Number of Participants
1750 participating women and 1100 partners (65%)
Study Centre
ErasmusMC, Rotterdam
Start time, Duration
Expected start date winter 2023, 2 years are needed for inclusion.
Interested in our Trial Programme?
Corsano Cardiowatch Bracelets enable continuous monitoring with multiple algorithms. Corsano is working closely with cardiologists, scientists, hospitals, patients, and research organisations. Scientific research demonstrates the legitimacy of Cardiowatch 287 algorithms.
We are currently performing pilots with selected clients. Contact us if you want to know more!
The founders of Corsano Health have over 100 years of experience in the Swiss Watch industry, with deep experience about ergonomic design and materials for wearables that are worn 24/7.