Title
Validation of Respiratory Rate with the Corsano Cardiowatch 287 Bracelet compared with Polygraphy
Topic
Evaluation of the performance and efficacy of the Corsano Cardiowatch 287 Bracelet in measuring respiratory rate at rest, as compared to golden standard and predicate device.
Number of Participants
25 subjects (at least 30% of each gender)
Inclusion / Exclusion criteria
Inclusion criteria:
People ≥ 18 years old
Exclusion criteria:
Wearer of cardiac implanted electronic device (Pacemaker, ICD)
CardioWatch 287 cannot be worn due to comprehensible reasons (allergic reactions, wounds, amputations, other)
Unable or not willing to sign informed consent
Significant mental or cognitive impairment
Currently enrolled in another clinical investigation in which the intervention might compromise the safety of the subject's participation in this study
Study Design
The patients are selected from the patient population referred for an overnight Polygraphy exam for a suspicion of sleep-disordered breathing. Patients are scheduled for a Respiratory Rate measurement, independent of the proposed study. Patients that are scheduled for these examinations and meet the inclusion criteria (see below) are approached by their practitioner or specialized nurse supervised by the for participation in the trial.
Target Points
The main study endpoints are the Percentage of respiratory rates of matching the resting Polygraphy recording. Percentage of respiration rate at rest ±2 BPM (beats per minute) matching the polygraphy device and manual counting.
Measurement procedures
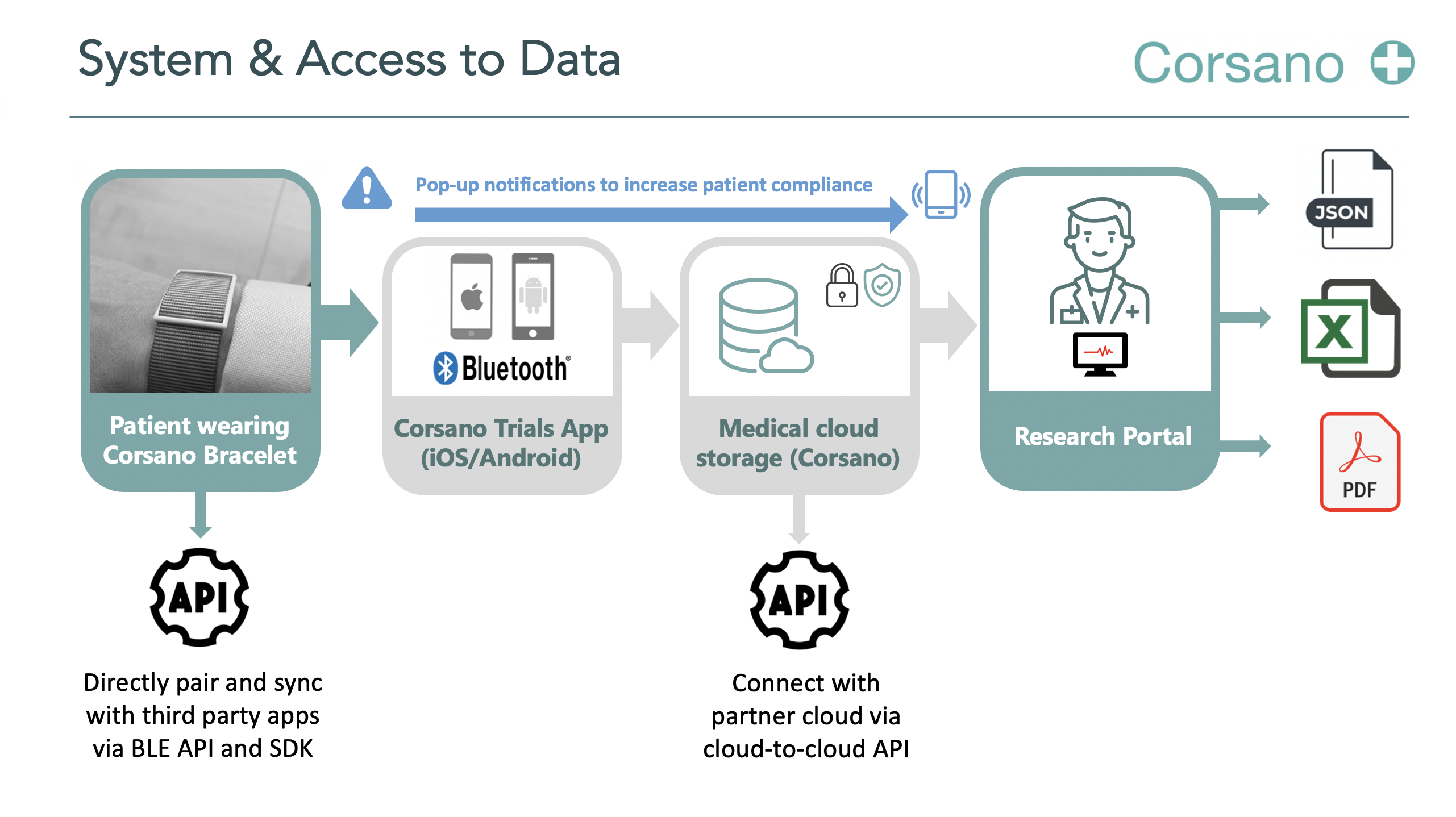
Participants are requested to wear a smartwatch or bracelet in addition to a Polygraph. Simultaneously, they will have the connected smartphone with them.
Study Centre
Haaglanden Clinics (NL)
Start time, Duration
November 2020, 6 months
Interested in our Trial Programme?
Corsano Cardiowatch Bracelets enable continuous monitoring with multiple algorithms. Corsano is working closely with cardiologists, scientists, hospitals, patients, and research organisations. Scientific research demonstrates the legitimacy of Cardiowatch 287 algorithms.
We are currently performing pilots with selected clients. Contact us if you want to know more!
The founders of Corsano Health have over 100 years of experience in the Swiss Watch industry, with deep experience about ergonomic design and materials for wearables that are worn 24/7.