Title
Photoplethysmography to detect circulatory arrest: A study in patients with induced cardiac arrests
Rationale
Out-of-hospital cardiac arrest (OHCA) is a leading cause of global mortality. Delays to initiation of cardiopulmonary resuscitation (CPR) should be as short as possible to achieve the highest survival chance. To reduce treatment delays automated cardiac arrest detection and call for help would be ideal. Photoplethysmography (PPG) is an optical technique often used in sport/smartwatches to monitor heart rhythm at the skin and based on previous studies it seems to have potential to detect circulatory arrest.
Objective
To study photoplethysmography signals during 1) induced circulatory arrest; 2) spontaneous circulatory arrest; 3) cardiac rhythms without circulatory arrest, and to construct a PPG-based algorithm for circulatory arrest detection.
Study design
Prospective multicenter observational study.
Study population
1) Patients undergoing defibrillation testing after (subcutaneous) implantable cardioverter defibrillator (ICD) implantation; 2) Patients undergoing rapid ventricular pacing during transcatheter aortic valve replacement (TAVR); 3) Patients undergoing ventricular tachycardia ablation; 4) Post-cardiac arrest patients.
Intervention
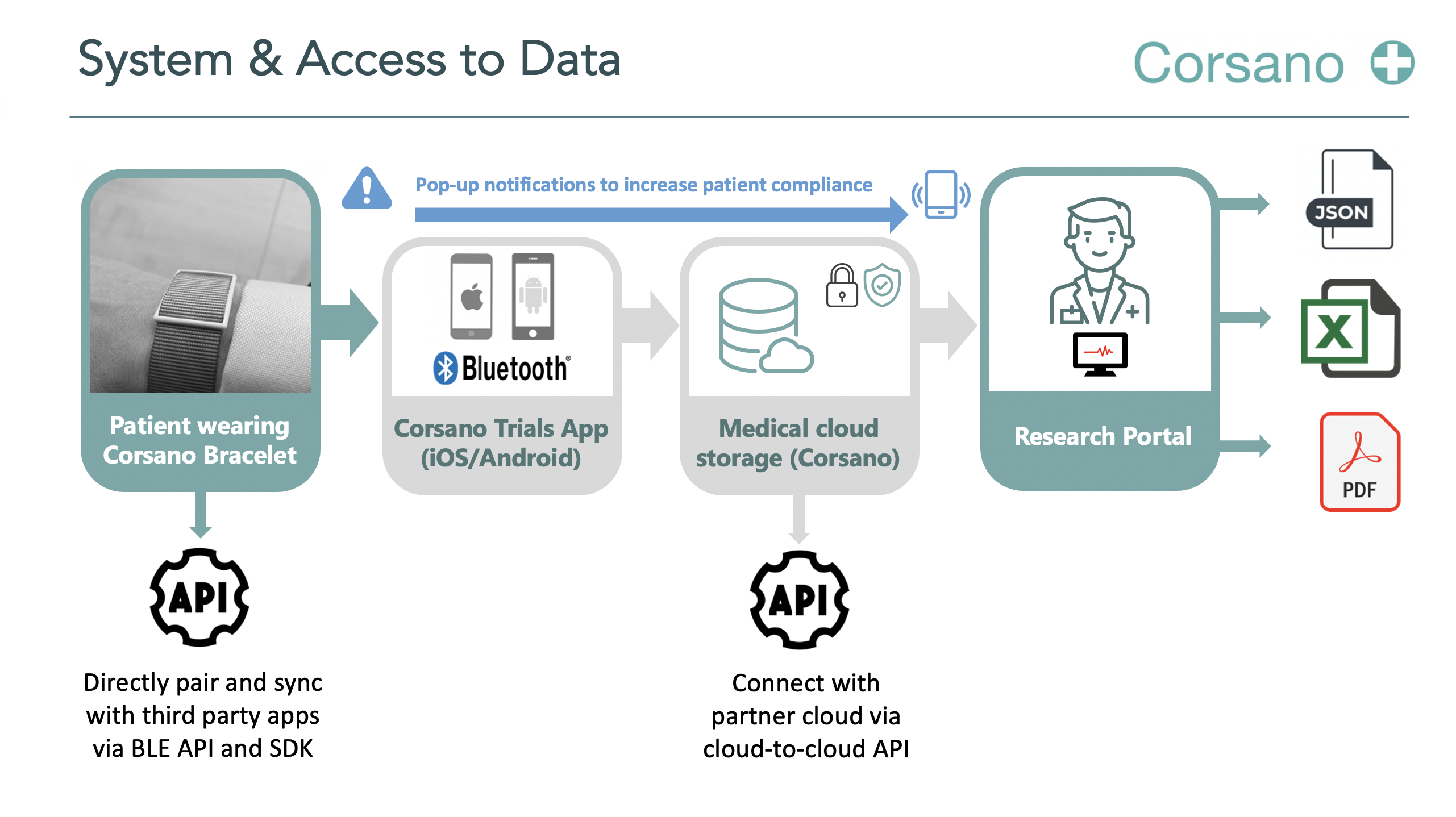
Patients will be equipped with Corsano Cardiowatch 287-2 to record PPG signals during hospital stay including during induced/spontaneous circulatory arrests. The circulatory arrest inductions are part of routine practice during above-described procedures and are not a study-related intervention.
Main study parameters/endpoints
Primary endpoints: Sensitivity and specificity for circulatory arrest. Secondary endpoints: Positive and negative predictive values for circulatory arrest, PPG-signal characteristics.
Nature and extent of the burden and risks associated with participation, benefit and group relatedness
Given the observational nature of this study and the non-invasive comfortable wristband used for PPG signal recording, the medical risk associated with study participation is negligible. There might be potential psychological impact of study participation for post-cardiac arrest patients, which may include settings involving re-arrests or palliative care settings.
Number of Participants
280 patients consisting of an estimated number of n=105 TAVR-patients (event rate 1/1), n=80 ICD-patients (event rate 1/1), n=10 VT-ablation patients (event rate 1/3) and n=60 post-cardiac arrest patients (event rate 1/5). Patients in whom ventricular fibrillation induction or rapid ventricular pacing does not result in complete circulatory arrest (as determined on (invasive) blood pressure measurements) will be replaced by new subjects to guarantee the anticipatednumber of true events.
Inclusion / Exclusion criteria
Inclusion criteria:
Undergoing ventricular fibrillation induction during defibrillation testing after ICD implantation
Undergoing rapid ventricular pacing during TAVR procedure
Undergoing ventricular tachycardia ablation
Post-cardiac arrest patients
Additionally, a subject must meet all of the following criteria:- Age => 18 years- Fitting the wristband
Exclusion criteria:
Unwilling or unable to provide informed consent
Known hemodynamically relevant subclavian artery stenosis
Medical issues that interfere with wearing of the wristband (e.g. skin disorders)
Unavailability of wristband used for PPG recording
Study Centres
RadboudMC, ErasmusMC, LUMC, Isala
Start time, Duration
November 2021. Expected study duration is 2 years
Interested in our Trial Programme?
Corsano Cardiowatch Bracelets enable continuous monitoring with multiple algorithms. Corsano is working closely with cardiologists, scientists, hospitals, patients, and research organisations. Scientific research demonstrates the legitimacy of Cardiowatch 287 algorithms.
We are currently performing pilots with selected clients. Contact us if you want to know more!
The founders of Corsano Health have over 100 years of experience in the Swiss Watch industry, with deep experience about ergonomic design and materials for wearables that are worn 24/7.